
Cary Blair Medium Rationale, Preparation and Uses
The half Cary Blair is a semi-solid agar, used for the transport and conservation of biological samples that harbor intestinal pathogens, labile and anaerobic microorganisms. Like any means of transport, its function is to keep the sample in optimal conditions until it is cultivated. The pathogenic microorganisms present, as well as the accompanying microbiota, must remain viable, but without increasing their population.
Cary Blair Medium is the result of the formulation change of Stuart Transport Medium. The modification was carried out in 1964 and consisted of replacing the pH regulating system (glycerophosphate) with a phosphated inorganic buffer..
The reformulation was necessary because its creators realized that glycerophosphate after a certain time could be metabolized by some saprophytic bacteria. These multiply overlap the pathogens present.
Other changes were the elimination of methylene blue, as well as a readjustment of the pH towards alkalinity (pH 8.4). All these changes improved the efficiency of the means of transport.
Originally the Cary Blair medium was used for the maintenance of intestinal pathogens, such as Shigella flexneri, Salmonella sp, Vibrio cholerae and Campylobacter sp.
However, the efficacy in preserving the viability of demanding and labile pathogenic microorganisms has subsequently been seen, such as: Neisseria gonorrhoeae, Haemophilus influenzae, Neisseria meningitidis, Bordetella pertussis, Streptococcus pneumoniae and some anaerobes.
Article index
- 1 Rationale
- 2 Preparation
- 3 Use
- 3.1 Type of samples
- 3.2 Sample collection
- 3.3 Identification of the sample
- 3.4 Transfer
- 4 Investigations carried out on the means of transport Cary Blair
- 5 Quality control
- 6 References
Basis
Cary Blair Medium contains Sodium Chloride, Sodium Thioglycolate, Disodium Phosphate, Calcium Chloride, and Agar.
Sodium chloride maintains the osmotic balance of the medium, disodium phosphate and calcium chloride balance the pH. For its part, sodium thioglycollate maintains a low oxidation-reduction potential, while the small portion of agar provides the semi-solid consistency..
Cary Blair medium does not contain nutritive substances, since the function of a transport medium is to keep the sample without it being modified in terms of humidity and microbial load; that is, it avoids dehydration of the sample while preserving the viability and quantity of the microorganisms present.
Finally, the slightly alkaline pH prevents the death of microorganisms by acidity, especially Vibrios are very sensitive to acids.
Preparation
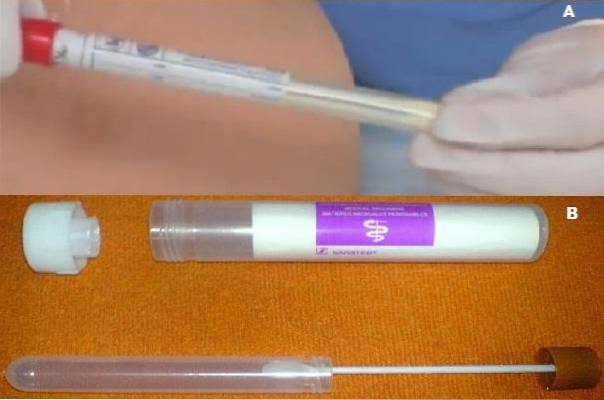
Cary Blair media is commercially available as a special ready-to-use device. The transport system is covered by a plastic Peel Pack bag. Inside it contains a swab for collecting the sample and the container tube for Cary Blair's semi-solid medium..
Also in the market there are commercial houses that supply the dehydrated medium for its preparation in the laboratory..
To do this, weigh 13.2 g of the dehydrated medium and dissolve it in one liter of distilled water. Heat and shake the preparation until completely dissolved. Distribute the medium into screw cap test tubes (Bakelite).
Steam the tubes for 15 minutes. Let cool and keep at room temperature until use.
Uninoculated Cary Blair medium can be stored optimally for up to 18 months at room temperature.
The pH of the medium must be adjusted to 8.4 ± 0.2. The color of the prepared medium is beige, with some opalescence.
Use
Type of samples
Cary Blair Medium is used when stool, rectal swab, and vaginal swab specimens cannot be processed immediately.
When no means of transport is available, samples can be stored at -70 ° C.
Sampling
The swab is impregnated with the sample and it is introduced into the Cary Blair medium, without reaching the bottom.
Sample identification
The Cary Blair medium must have a label in which the basic data are placed, such as: first and last name of the patient, identification number of the sample, source of the sample collection, treating physician, date and time of the collection. sample, presumptive diagnosis, antibiotic treatment.
Transfer
Tightly covered and transported at room temperature for fecal samples. Whereas, for vaginal samples in search of anaerobic microorganisms, it is recommended to transport the samples at 4 ° C.
In all cases, the recommended transfer time is 4-6 hours, with a maximum of 24 hours..
If the presence of Campylobacter jejuni in stool samples and these cannot be processed within 24 hours, storage in Cary Blair medium at 4 ° C is recommended.
Investigations carried out on the Cary Blair means of transport
Several studies have shown that Cary Blair medium can remain viable to enteropathogenic microorganisms of the genus Shigella and Salmonella for up to 49 days after taking the sample..
For his part, Vibrio cholerae, another important intestinal pathogen, is capable of surviving for 22 days, while Yersinia pestis (causative agent of bubonic and pneumonic plague) can be recovered after 75 days.
However, despite the demonstrated durability in terms of the recovery of these microorganisms, it is recommended that the samples taken be transported in the Cary Blair medium to the laboratory as quickly as possible..
Likewise, once the sample reaches the laboratory, it must be sown without delay in the respective culture media..
On the other hand, the Cary Blair medium, in addition to being useful for the transport of facultative microorganisms, is also recommended for samples containing anaerobic pathogens..
In this sense, DeMarco and collaborators in 2017 carried out a study entitled: Survival of vaginal microorganisms in three commercially available transport systems.
They demonstrated that the Cary Blair transport medium is the best transport medium for the preservation and recovery of vaginal anaerobic microorganisms. Likewise, they showed that the highest recovery rate was achieved when the transfer temperature was 4 ° C..
Therefore, it is concluded that the transport temperature for vaginal swabs (anaerobic microorganisms) should be at 4 ° C. While for fecal samples looking for facultative microorganisms, the ideal temperature is room temperature..
QA
Strains known as Shigella sonnei ATCC 11060, Salmonella choleraesuis ATCC 14028, Vibrio cholerae Y Campylobacter sp.
The medium is inoculated with the chosen strain and kept at room temperature for 24 hours. They are then sown in the corresponding culture media. In all cases, a satisfactory recovery of the microorganism in question is expected..
References
- DeMarco AL, Rabe LK, Austin MN, et al. Survival of vaginal microorganisms in three commercially available transport systems. Anaerobe. 2017; 45: 44-49.
- Wasfy M, Oyofo B, Elgindy A, Churilla A. Comparison of preservation media for storage of stool samples. J Clin Microbiol. nineteen ninety five; 33 (8): 2176-2178.
- Dan M, Richardson J, Miliotis MD, Koornhof HJ. Comparison of preservation media and freezing conditions for storage of specimens of faeces. J Med Microbiol. 1989; 28 (2): 151-4.
- Koneman E, Allen S, Janda W, Schreckenberger P, Winn W. (2004). Microbiological Diagnosis. 5th ed. Editorial Panamericana S.A. Argentina.
- Forbes B, Sahm D, Weissfeld A. (2009). Bailey & Scott Microbiological Diagnosis. 12 ed. Editorial Panamericana S.A. Argentina.
- Conda Pronadisa Laboratories. Half Cary Blair. Available at: condalab.com
- Metrix Laboratory. Cary Blair. Available at: metrixlab.mx



Yet No Comments