
Distilled water properties, how to make it, uses and toxicity
The distilled water It is a type of water purified by the distillation procedure, which leaves it free of organic impurities and ions. This is produced when tap water is boiled, the vapors of which are transported and condensed by a condenser. This is how distilled water is obtained.
The impurities in the tap water remain in the original container, and by condensation water is obtained without the non-volatile solids. The distillation process removes many impurities present in common water.
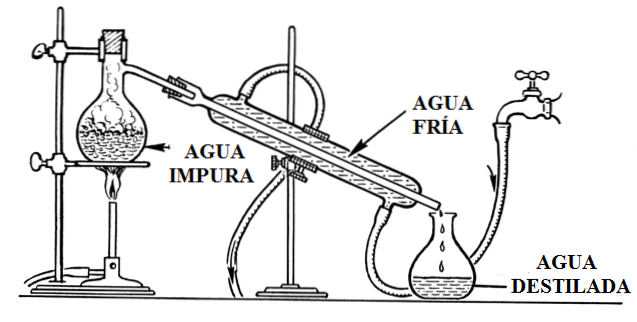
A typical distillation setup is shown in the image above. It illustrates everything that has been said above. Cold water runs through the hoses, which absorbs the heat of the aqueous vapors, condensing and sliding through the interior of the condenser to the collection container..
The distillation of water can be obtained with the proper assembly of the materials shown in the figure below. This assembly can be reproduced, with different pieces, in any laboratory.
However, there is a great variety of water distillation equipment, in addition to the rudimentary assembly, whose distillation performance in liters per hour is higher. There are different commercial brands, automatic distillers of continuous production, among others..
It is important to store distilled water in containers or containers that are subjected to exhaustive cleaning and disinfection with some regularity; like for example, the pizetas.
There are numerous procedures that are carried out at a scientific, industrial and commercial level that require the use of distilled water. The use of tap water or water that has not been purified by distillation would cause interference or errors in many chemical reactions and procedures..
For example, the ions found in common water have a corrosive effect on vehicle engines, reducing their useful life..
Article index
- 1 Properties of distilled water
- 2 How to make distilled water in the laboratory?
- 2.1 Vaporization and condensation
- 2.2 Distillation equipment
- 3 Uses
- 3.1 In research
- 3.2 Rinsing of laboratory material and equipment
- 3.3 In everyday appliances
- 3.4 At an industrial level
- 3.5 In the health sector
- 4 Toxicity
- 5 References
Properties of distilled water
The properties of distilled water are very similar to those described for water in general, untreated. However, among the properties that differentiate distilled water from normal water is its electrical conductivity, which is almost nil, due to the lack of ions; practically non-existent in double distilled water.
Distilled water is a type of treated water that lacks ions, such as the abundant chlorine in tap water. In addition, it is free of impurities, dissolved substances, microorganisms, and other elements such as mud and earth..
It differs from deionized (or demineralized) water, in that its production process is distillation; while for deionized water, it is obtained through the use of ion exchangers, which cannot remove organic matter.
In this sense, a property of distilled water is also its low content of microorganisms. From this point of view, it is less dangerous to ingest distilled water than deionized water..
How to make distilled water in the laboratory?
Using the distillation method, common water is purified, colloquially called jet, tap, running or potable water. The most important processes carried out to obtain or generate distilled water are vaporization and condensation..
The operation of the distillation equipment was based on the principle that exists in nature in the water cycle.
Vaporization and condensation
The vaporization process is achieved by heating tap water, increasing its temperature until it reaches its boiling point. At the appropriate temperature and pressure, the water boils and evaporates, going from the liquid state to the gaseous state.
Some heat sources used to heat the water can be a lighter, or electric resistances that supply the thermal energy (heating blankets).
With the flame of the lighter the first container or balloon containing the running water with impurities is heated. When water boils, the steam passes into the internal part of a condenser which is cooled by receiving jet water.
Condensation occurs when this water vapor cools, condenses and passes from a gaseous state to a liquid. The surroundings of the central conduit of the condenser through which the steam circulates is cooled by the flow of water.
The gradually generated distilled water is collected and stored in a different container, tank or drum free of impurities. Substances that are mixed or dissolved in water do not evaporate, and therefore remain in the container of tap water.
In this way, ions and organic impurities are left behind in the initial container..
Distillation equipment
There are diversity of water distillation equipment. They can be manual, simple (as shown in the image), complex, or automatic. There are tabletop stills, stills that can be hung on the wall, or that come with their own cabinet..
Its capacity and design depend on the demand or the amount of distilled water required. The simplest water distillation equipment or apparatus that can be used in laboratories is known as the alembic (image below).

There are different types of water stills, manual or automatic process, continuous or discontinuous. The highlight is that the basis of its operation is the same: based on the processes of vaporization and condensation..
Applications
In research
-Distilled water is used in research, teaching and routine laboratories.
-The reagent solutions, as well as their continuous dilutions, are made with distilled water..
-The use of distilled water is required in numerous analytical processes carried out in chemistry and biology laboratories..
Laboratory equipment and material rinsing
-It is used very frequently in the rinse or last step of washing laboratory equipment. It is available in pizetas to rinse glass material such as cylinders, pipettes, test tubes, spouts or tips of automatic pipettes, reaction plates, etc..
-A concrete example of the use of distilled water is in the washing or rinsing of delicate equipment, such as the pH meter. When conducting pH titrations, the electrodes should be thoroughly rinsed or washed with distilled water after use. Thus, ions that may interfere with future readings are eliminated..
-Double distilled water, obtained from two consecutive distillation processes, is used in some chemical procedures that require water of optimum purity; for example, in the preparation of aqueous mobile phases in high performance liquid chromatography.
In everyday appliances
-The use of distilled water increases the life of batteries and vehicle cooling systems. It is recommended to use it on plates that use steam, to reduce metal corrosion. It is also used in humidifiers, among other laboratory equipment that need water.
-Distilled water is used to clean equipment with screens, monitors. Does not leave white spots on its surface.
-In aquarium waters, although there is controversy, since fish need the minerals that raw water has. On the other hand, distilled water has a lower concentration of organic matter; that is, bacteria and viruses, which can be harmful to fish.
At an industrial level
-Distilled water is used in the production of beverages such as beer, in canned vegetables and fruits, among others..
-The use of distilled water is essential in the pharmaceutical, cosmetic and food industries, among other areas..
-There are cases in which it is necessary to treat sea water; large vessels consume distilled water from sea water. The same happens when the water is not sweet for human consumption: it is purified by distillation before use.
In the health sector
-It is important to use distilled water in routine and special analysis laboratories..
-It is required for the preparation of reagents, in the materials washing and sterilization units, and also in the dietetic and pharmacy units.
-In the preparation of treatments, for example, medications that require it are diluted with distilled water.
Toxicity
In general, the production of toxic effects of distilled water on living beings is not described. According to toxicological information, distilled water as a product is not classified as dangerous. There are also no references that indicate that distilled water affects the environment; that is, it lacks ecotoxicity.
There are no harmful effects described if distilled water is inhaled, ingested or comes into contact with mucous membranes or the skin. Similarly, when handling it, it is not necessary to use barriers such as gloves, glasses, or protective equipment..
However, the consumption of distilled water is not recommended, since it does not provide any of the minerals that the body needs. On the contrary, when ingested in large quantities, it can have a demineralizing effect on the body, facilitating the elimination of ions in the urine..
Contrary to what happens in the human organism or body, with plants the situation is different. It is ideal to water the plants with distilled water because it lacks chlorine; since the minerals they need are taken from the earth through their roots and by capillarity.
References
- All About Water Filters. (2019). 13 different uses of distilled water. Recovered from: all-about-water-filters.com
- Distilled Water (2012). Material Safety Data Sheet. Recovered from: ehs.cranesville.com
- H2OLabs. (2019). Water Distiller From H2O Labs. Recovered from: www.h2olabs.com
- Wikipedia. (2019). Distilled water. Recovered from: en.wikipedia.org
- Pure water. (2017). Distilled or deionized water is there a difference? Which is better? Recovered from: aguapuraysana.com



Yet No Comments