
Aspergillus terreus taxonomy, morphology and life cycle
Aspergillus terreus It is a kind of fungus that produces secondary metabolites such as patulin, citrinin, and glyotoxins, which are harmful to humans. It is known for its refractance to amphotericin B therapy. It may be an opportunistic pathogen causing invasive pulmonary aspergillosis in immunosuppressive patients..
A. terreus is also used to metabolize "lovastatin", a compound used in the pharmaceutical industry to regulate cholesterol levels. It also produces beneficial secondary metabolites such as terrein, an inhibitor of melanogenesis, asperfuranone and cyclosporin A, which are used as immunosuppressive drugs..
Even some strains are used for the production of organic acids, itaconic acids and itatartaric acids through fermentation processes..
Article index
- 1 Taxonomic identification of A. terreus
- 2 Morphology
- 2.1 Macroscopically
- 2.2 Microscopically
- 3 Life cycle
- 4 References
Taxonomic identification of A. terreus
The genus Aspergillus, to which A. terreus belongs, has undergone extensive taxonomic studies based on its genomic DNA. Many of these studies have focused on specific groups (species, section and subgenus).
A. terreus belongs to the subgenus Nidulantes of the Terrei section. With advances in molecular biology studies, it has been recognized that there is genetic variability that can distinguish strains of the same species by protein patterns.
Morphology
Morphologically A. terreus is a filamentous fungus as are the species of the genus Aspergillus.
Macroscopically
Macroscopically, the fungus can be characterized on specialized culture media or on the substrates where it grows. A culture medium used in the laboratory to plant the fungus is CYA medium (Yeast extract and Czapek agar) and MEA medium (Malt extract agar), allowing the observation of the colony, color, diameter and even the formation of structures. reproduction or resistance, depending on conditions and incubation time.
A. terreus, on CYA medium, is observed as a circular colony (30-65 mm in diameter) with a velvety or woolly texture, flat or with radial furrows, with white mycelium..
The color can vary from cinnamon brown to yellowish brown, but when looking at the back of the culture plate, it can be seen as yellow, gold or brown and sometimes with a yellow diffusible pigment in the middle..
If the medium is MEA, the colonies are sparse, flesh-colored or pale orange to orange-gray, with barely visible white mycelium. When looking at the back of the plate, the colonies are seen with yellowish tones.
Microscopically
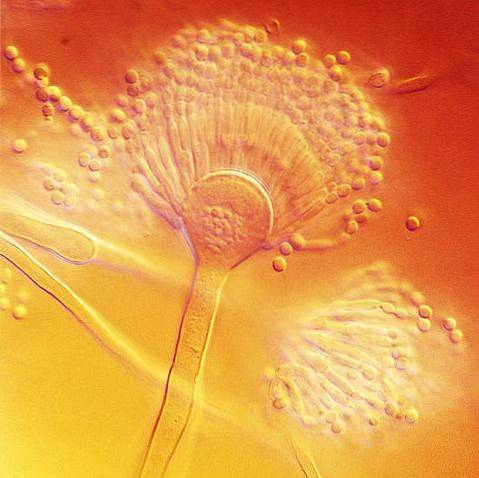
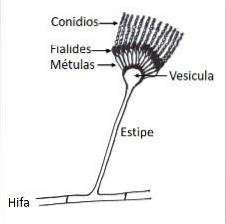
Microscopically, like all species of the genus Aspergillus, it has specialized hyphae called conidiophores, on which the conidiogenous cells that form the conidia or asexual spores of the fungus will develop..
The conidiophore is formed by three well differentiated structures; the vesicle, the stipe and the foot cell that links with the rest of the hyphae. Conidiogenous cells, called phialides, will form on the vesicle, and depending on the species other cells develop between the vesicles and the phialides, called métulas..
A. terreus forms conidiophores with conidial heads in compact columns, with spherical or subglobose vesicles, measuring 12-20 µm in width. The stipe is hyaline and can vary in length from 100-250 µm.
It has metulas (what is known as biserial conidial heads) with dimensions ranging from 5-7 µm x 2-3 µm and phialides of 7 µm x 1.5 - 2.5 µm. The smooth, globose or subglobose conidia are small compared to other Aspergillus species and can measure 2 -2.5 µm.
With advances in molecular biology and sequencing techniques, today the identification of fungal species is facilitated by the use of molecular markers that allow the study of strains of a species. Currently the barcode of many fungi is the spacer regions of ribosomal DNA.
Biological cycle
A sexual phase and an asexual phase can be identified. When a spore reaches the ideal substrate, a phase of approximately 20 hours is required for the hyphae to develop.
If conditions are favorable, such as good aeration and sunlight, the hyphae begin to differentiate, thickening a part of the cell wall from which the conidiophore will emerge..
This will develop the conidia that will be scattered by the wind, restarting the life cycle of the fungus. If conditions are not favorable for vegetative development, such as long hours of darkness, the sexual phase of the fungus can develop.
In the sexual phase, cell primordia develop that give rise to a globose structure called cleistothecia. Inside are the asci where the ascospores will develop. These are the spores that under favorable conditions and on a suitable substrate will develop hyphae, restarting the life cycle of the fungus.
References
- Samson RA, Visagie CM, Houbraken J., Hong S.-B., Hubka V., Klaassen CHW, Perrone G., Seifert KA, Susca A., Tanney JB, Varga J., Kocsub S., Szigeti G., Yaguchi T., and Frisvad JC… 2014. Phylogeny, Identification and nomenclature of the genus Aspergillus. Studys in Mycology 78: 141-173.
- It covers Mª L. 2000. Taxonomy and identification of species involved in nosocomial aspergillosis. Rev Iberoam Micol 2000; 17: S79-S84.
- Hee-Soo P., Sang-Cheol J., Kap-Hoon H., Seung-Beom H., and Jae-Hyuk Y. 2017. Chapter Three. Diversity, applications and synthetic biology of industrially important Aspergillus fungi. Advances in Microbiology 100: 161-201.
- Rodrigues A.C. 2016. Chapter 6. Secondary metabolism and antimicrobial metabolites of Aspergillus. In: New and Future Developments in Microbial Biotechnology and Bioengineering. P 81-90.
- Samson RA, Visagie CM, Houbraken S., Hong B., Hubka V., Klaassen CHW, Perrone G., Seifert KA, Susca A., Tanney JB, Verga J., Kocsubé S., Szigeti G., Yaguchi T. and Frisvad JC 2014. Phylogeny, identification and nomenclature of the genus Aspergillus. Studies in Mycology 78: 141-173.
- Arunmonzhi B. S. 2009. Aspergillus terreus complex. Medical Mycology 47: (Supplement 1), S42-S46.
- Narasimhan B. and Madhivathani A. 2010. Genetic variability of Aspergillus terreus from dried grapes using RAPD-PCR. Advances in Bioscience and Biotechnology 1: 345-353 ABB.
- Bayram Ö., Braus G. H., Fischer R. and Rodriguez-Romero J. 2010. Review Spotlight on Aspergillus nidulans photosensory systems. Fungal Genetics and Biology 47: 900-908.



Yet No Comments