
Sugars Reductures Methods For Determination, Importance
The reducing sugars they are biomolecules that function as reducing agents; that is, they can donate electrons to another molecule with which they react. In other words, a reducing sugar is a carbohydrate that contains a carbonyl group (C = O) in its structure..
This carbonyl group is made up of a carbon atom bonded to an oxygen atom through a double bond. This group can be found in different positions in sugar molecules, resulting in other functional groups such as aldehydes and ketones..
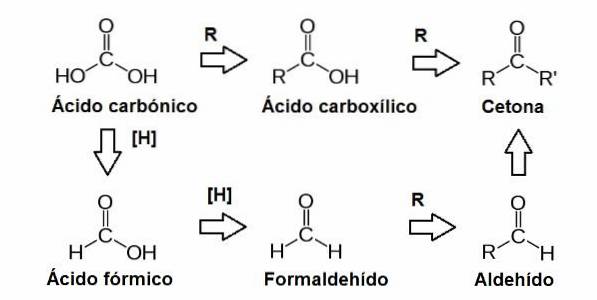
Aldehydes and ketones are found in the molecules of simple sugars or monosaccharides. These sugars are classified into ketoses if they have the carbonyl group inside the molecule (ketone), or aldoses if they contain it in the terminal position (aldehyde)..
Aldehydes are functional groups that can carry out oxidation-reduction reactions, which involve the movement of electrons between molecules. Oxidation occurs when a molecule loses one or more electrons, and reduction occurs when a molecule gains one or more electrons..
Of the types of carbohydrates that exist, monosaccharides are all reducing sugars. For example, glucose, galactose, and fructose function as reducing agents..
In some cases, monosaccharides are part of larger molecules such as disaccharides and polysaccharides. For this reason, some disaccharides -such as maltose- also behave as reducing sugars..
Article index
- 1 Methods for the determination of reducing sugars
- 1.1 Benedict's test
- 1.2 Fehling's reagent
- 1.3 The Tollens reagent
- 2 Importance
- 2.1 Importance in medicine
- 2.2 The Maillard reaction
- 2.3 Food quality
- 3 Difference between reducing sugars and non-reducing sugars
- 4 References
Methods for the determination of reducing sugars
Benedict's test
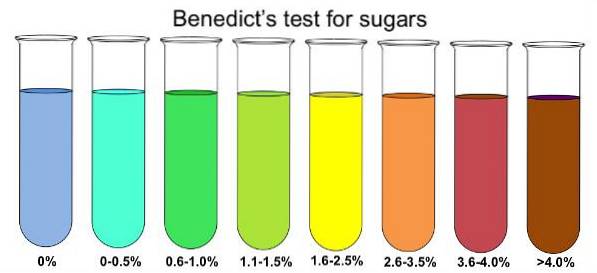
To determine the presence of reducing sugars in a sample, it is dissolved in boiling water. Next, add a small amount of Benedict's reagent and wait for the solution to reach room temperature. Within 10 minutes the solution should begin to change color.
If the color changes to blue, then there are no reducing sugars present, particularly glucose. If there is a large amount of glucose present in the sample to be tested, then the color change will progress to green, yellow, orange, red, and finally brown..
Benedict's reagent is a mixture of several compounds: it includes anhydrous sodium carbonate, sodium citrate, and copper (II) sulfate pentahydrate. Once added to the solution with the sample, the possible oxidation-reduction reactions will begin.
If reducing sugars are present, they will reduce the copper sulfate (blue color) in the Benedict solution to a copper sulfide (reddish color), which looks like the precipitate and is responsible for the color change..
Non-reducing sugars cannot do this. This particular test only provides a qualitative understanding of the presence of reducing sugars; that is, it indicates whether or not there are reducing sugars in the sample.
Fehling's reagent
Similar to the Benedict test, the Fehling test requires that the sample be completely dissolved in a solution; this is done in the presence of heat to ensure that it dissolves completely. After this, the Fehling solution is added, stirring constantly.
If reducing sugars are present, the solution should begin to change color as an oxide or red precipitate forms. If no reducing sugars are present, the solution will remain blue or green. Fehling's solution is also prepared from two other solutions (A and B).
Solution A contains copper (II) sulfate pentahydrate dissolved in water, and solution B contains sodium potassium tartrate tetrahydrate (Rochelle's salt) and sodium hydroxide in water. The two solutions are mixed in equal parts to make the final test solution..
This test is used to determine monosaccharides, specifically aldoses and ketoses. These are detected when the aldehyde oxidizes to acid and forms a cuprous oxide.
Upon contact with an aldehyde group, it is reduced to a cuprous ion, which forms the red precipitate and indicates the presence of reducing sugars. If there were no reducing sugars in the sample, the solution would remain blue, indicating a negative result for this test..
Tollens reagent
The Tollens test, also known as the silver mirror test, is a qualitative laboratory test used to distinguish between an aldehyde and a ketone. Exploits the fact that aldehydes are easily oxidized, whereas ketones do not.
The Tollens test uses a mixture known as Tollens reagent, which is a basic solution containing silver ions coordinated with ammonia..
This reagent is not commercially available due to its short shelf life, so it must be prepared in the laboratory when it is to be used..
Reagent preparation involves two steps:
Step 1
Aqueous silver nitrate is mixed with aqueous sodium hydroxide.
Step 2
Aqueous ammonia is added dropwise until the precipitated silver oxide is completely dissolved..
The Tollens reagent oxidizes the aldehydes that are present in the corresponding reducing sugars. The same reaction involves the reduction of silver ions from Tollens' reagent, which converts them to metallic silver. If the test is carried out in a clean test tube, a silver precipitate forms..
Thus, a positive result with the Tollens reagent is determined by observing a "silver mirror" inside the test tube; this mirror effect is characteristic of this reaction.
Importance
Determining the presence of reducing sugars in different samples is important in several aspects including medicine and gastronomy..
Importance in medicine
Tests for reducing sugars have been used for years to diagnose patients with diabetes. This can be done because this disease is characterized by an increase in blood glucose levels, with which the determination of these can be carried out by these oxidation methods.
By measuring the amount of oxidizing agent reduced by glucose, it is possible to determine the glucose concentration in blood or urine samples.
This allows the patient to be instructed on the appropriate amount of insulin to inject to bring blood glucose levels back into the normal range..
Maillard's reaction
The Maillard reaction includes a set of complex reactions that occur when cooking some foods. By increasing the temperature of food, the carbonyl groups of reducing sugars react with the amino groups of amino acids.
This cooking reaction generates various products and, although many are beneficial to health, others are toxic and even carcinogenic. For this reason, it is important to know the chemistry of the reducing sugars that are included in the normal diet..
When cooking foods rich in starch -such as potatoes- at very high temperatures (greater than 120 ° C), the Maillard reaction occurs.
This reaction occurs between the amino acid asparagine and reducing sugars, generating acrylamide molecules, which is a neurotoxin and a possible carcinogen..
Food quality
The quality of certain foods can be monitored using reducing sugar detection methods. For example: in wines, juices and sugar cane, the level of reducing sugars is determined as an indication of the quality of the product..
For the determination of reducing sugars in food, Fehling's reagent with methylene blue is normally used as an oxide-reduction indicator. This modification is commonly known as the Lane-Eynon method..
Difference between reducing sugars and non-reducing sugars
The difference between reducing and non-reducing sugars is in their molecular structure. Carbohydrates that other molecules reduce do so by donating electrons from their free aldehyde or ketone groups.
Therefore, non-reducing sugars do not have free aldehydes or ketones in their structure. Consequently, they give negative results in the tests for the detection of reducing sugars, such as the Fehling or Benedict tests..
Reducing sugars comprise all monosaccharides and some disaccharides, while non-reducing sugars include some disaccharides and all polysaccharides.
References
- Benedict, R. (1907). THE DETECTION AND ESTIMATION OF REDUCING SUGARS. Journal of Biological Chemistry, 3, 101-117.
- Berg, J., Tymoczko, J., Gatto, G. & Strayer, L. (2015). Biochemistry (8th ed.). W. H. Freeman and Company.
- Chitvoranund, N., Jiemsirilers, S., & Kashima, D. P. (2013). Effects of Surface Treatment on Adhesion of Silver Film on Glass Substrate Fabricated by Electroless Plating. Journal of the Australian Ceramic Society, 49(1), 62-69.
- Hildreth, A., Brown, G. (1942). Modification of Lane-Eynon method for sugar determination. Journal Association of Official Analytical Chemists 25 (3): 775-778.
- Jiang, Z., Wang, L., Wu, W., & Wang, Y. (2013). Biological activities and physicochemical properties of Maillard reaction products in sugar-bovine casein peptide model systems. Food Chemistry, 141(4), 3837-3845.
- Nelson, D., Cox, M. & Lehninger, A. (2013). Lehninger Principles of Biochemistry (6th). W.H. Freeman and Company.
- Pedreschi, F., Mariotti, M. S., & Granby, K. (2014). Current issues in dietary acrylamide: Formation, mitigation and risk assessment. Journal of the Science of Food and Agriculture, 94(1), 9-20.
- Rajakylä, E., & Paloposki, M. (1983). Determination of sugars (and betaine) in molasses by high-performance liquid chromatography. Journal of Chromatography, 282, 595-602.
- Scales, F. (1915). THE DETERMINATION OF REDUCING SUGARS. The Journal of Ciological Chemistry, 2. 3, 81-87.
- Voet, D., Voet, J. & Pratt, C. (2016). Fundamentals of Biochemistry: Life at the Molecular Level(5th ed.). Wiley.



Yet No Comments