
Ethidium bromide structure, properties, uses, toxicity
The ethidium bromide It is a fluorescent and aromatic compound that has the ability, due to its chemical structure, to intercalate between DNA chains. It also binds to highly folded RNA molecules. This allows for an interaction between this salt and the nitrogenous bases..
Ethidium bromide absorbs ultraviolet light in a wavelength range from 210 nm to 285 nm, emitting an orange fluorescence of 605 nm. The intensity of its fluorescence increases up to 20 times when it interacts with DNA.
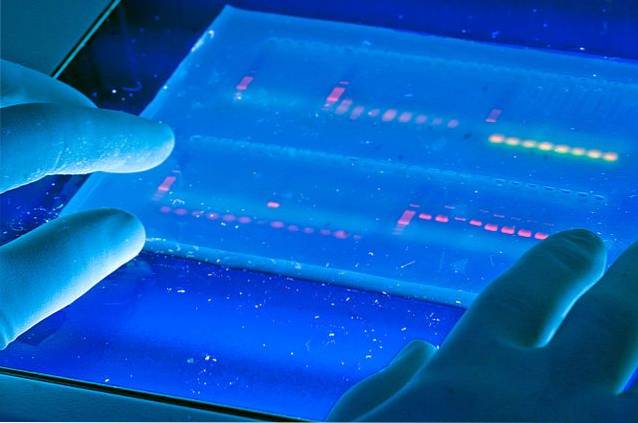
Due to its fluorescence property, ethidium bromide is used to visualize separated DNA fragments by agarose electrophoresis (upper image); technique introduced independently by Aaij and Borst (1972) and Sharp (1973).
Ethidium bromide, when intercalated between DNA chains, could also hinder its duplication and transcription processes; and therefore, be the cause of the generation of mutations. However, there is no conclusive evidence to support this assumption..
Article index
- 1 Chemical structure
- 2 Properties
- 2.1 Name
- 2.2 Molecular formula
- 2.3 Physical appearance
- 2.4 Flavor
- 2.5 Odor
- 2.6 Melting point
- 2.7 Flash point
- 2.8 Solubility
- 2.9 Density
- 2.10 Vapor pressure
- 2.11 Octanol / water partition coefficient
- 2.12 Decomposition
- 2.13 Stability
- 2.14 Refractive index
- 2.15 pH
- 3 Uses
- 3.1 Polymerase chain reaction (PCR)
- 3.2 Electrophoresis of DNA in agarose and acrylamide gel
- 3.3 Action of ethidium bromide on trypanosomes
- 3.4 Use in an animal model of multiple sclerosis
- 4 Toxicity
- 4.1 Exposure
- 4.2 Mutagenicity
- 5 References
Chemical structure

In the upper image we have the molecular structure of ethidium bromide represented by its structural formula.
The molecule is almost totally flat, because all the atoms of the system formed by the three rings (phenanthridine) and the positively charged nitrogen atom have sp hybridizationtwo. But it does not happen in the same way with their substituent groups.
The phenyl group at the extreme right, the amino groups, and the ethyl group attached to the charged nitrogen are responsible for the system absorbing the UV wavelengths that later characterize the fluorescence of the ethidium bromide..
On the other hand, note that their intermolecular interactions are mainly governed by electrostatic attractions; and to a lesser degree, they are held together by the London dispersive forces of the rings.
Properties
Name
Ethidium bromide.
IUPAC name: 3,8-Diamino-5-ethyl-6-phenylphenanthridinium bromide.
Synonyms: homide bromide and Dromilac.
Molecular formula
Ctwenty-oneHtwentyN3Br
Physical appearance
Dark red crystals or occurs as a brown powder.
Taste
Bitter.
Odor
Odorless solid.
Melting point
260 - 262 ºC (decomposes).
ignition point
> 100 ºC
Solubility
40 g / L at 25 ºC in water, and in ethanol 2 mg / mL.
Density
0.34 g / cm3
Vapor pressure
1,210-12 mmHg at 25 ° C (estimated).
Octanol / water partition coefficient
Log Kow = - 0.38
Decomposition
When it is heated to decomposition, ethidium bromide emits a very toxic smoke of hydrogen bromide and nitrogen oxides..
Stability
Stable and incompatible with strong oxidizing agents.
Refractive index
1.67 (estimated).
pH
4-7 in a 2% solution in water.
Applications
Polymerase chain reaction (PCR)
The polymerase chain reaction, PCR, makes it possible to obtain many copies in an exponential way starting from a DNA fragment. The technique is based on the property of the DNA polymerase enzyme to replicate DNA strands from its fragments that serve as a template..
It is a technique that has many applications, including the detection of mutations related to hereditary diseases; paternity tests; identification of a person who committed a crime, etc..
Ethidium bromide helps to identify DNA fragments, products of their enzymatic degradation that can be used in the PCR technique.
Acrylamide agarose gel electrophoresis of DNA
The ethidium bromide is incorporated into the gel prior to performing the electrophoresis. The compound is sandwiched between the DNA bands and produces a fluorescence when exposed to ultraviolet light that serves to reveal the DNA fragments that are separated in electrophoresis.
The fluorescence pattern of the electrophoresis serves as an orientation about the origin of the DNA fragments. The binding of ethidium bromide to DNA changes the conformation, charge, weight and flexibility of the DNA molecule, which results in a reduction in the mobility of the macromolecule..
This effect increases as the size of the DNA fragment increases..
Action of ethidium bromide on trypanosomes
Ethidium bromide began to be used in the treatment of trypanosomiasis in cattle in the 1950s, under the name Homidio. From there came the name homide bromide as a synonym for ethidium bromide.
The therapeutic use of ethidium bromide is based on its toxicity to the mitochondria. This is manifested by a reduction in the number of copies of mitochondrial DNA..
Ethidium bromide binds to the DNA molecules of the trypanosome kinetoplast and changes its conformation to DNAz. This form of DNA is lethal, since its replication is inhibited.
Use in an animal model of multiple sclerosis
Direct injection of ethidium bromide into the cisterna magna produced a reproducible acute myelin loss lesion in the brainstem of rats. The same spinal cord injection in cats produced an injury comparable to that seen in rats.
Multiple sclerosis is an autoimmune disease of the nervous system, in which the immune system destroys myelin, a substance that covers neurons.
Toxicity
Exposition
Ethidium bromide is considered a toxic compound, since by inhalation it causes acute irritation of the respiratory tract. Also, by contact with the skin, ethidium bromide can cause inflammation and / or discoloration..
Meanwhile, in the eyes, acute exposure causes irritation, redness and eye pain. Therefore, it is recommended that the material used with ethidium bromide be handled with the Material Safety Data Sheet (MSDS)..
Mutagenic character
Ethidium bromide is considered a highly mutagenic compound, since when intercalated in DNA it could affect its duplication and transcription, causing mutations; and even, a possible carcinogenic action has been pointed out.
The AMES Test only detected the induction of mutations by ethidium bromide in bacteria, when a liver homogenate was used in the test..
This led us to think that it is not directly responsible for the appearance of mutations in bacteria, but that these could be the consequence of the action of some metabolite generated in the interaction of ethidium bromide with the liver homogenate..
On the other hand, The Natural Toxicology Program established that ethidium bromide was not mutagenic for rats and mice. Despite this, its use is being reduced in the laboratories that use it in their research..
However, the concentration of ethidium bromide used in the investigations is one thousandth of the concentration of the dose given to cattle in the treatment of trypanosomiasis, without the report of the appearance of mutations..
References
- Doronina Vicki. (2017). Burning bright: a brief history of ethidium bromide DNA staining. Recovered from: bitesizebio.com
- Wikipedia. (2020). Ethidium bromide. Recovered from: en.wikipedia.org
- ElSevier B.V. (2020). Ethidium bromide. ScienceDirect. Recovered from: sciencedirect.com
- Royal Society of Chemistry. (2020). Ethidium bromide. Recovered from: chemspider.com
- Chemical Book. (2017). Ethidium bromide. Recovered from: chemicalbook.com
- Polytechnic university of Valencia. (2012). Standard procedure for work with ethidium bromide. Recovered from: sprl.upv.es



Yet No Comments