
Benzyl acid synthesis, rearrangement and uses

The benzyl acid, Also known as diphenylglycolic acid or 2-hydroxy-2,2-diphenylacetic acid, it is an aromatic organic compound that is used in the synthesis of compounds with different utilities. It is a 2-hydroxy monocarboxylic acid, whose chemical formula is C14H12O3 (it has two phenolic rings in its structure) and has a molecular weight of 228.25 g / mol.
It is normally used in the chemical industry, where it is acquired as a yellowish crystalline powder with good solubility in primary alcohols and with a boiling point close to 180 ° C..

It has been classified as a "Brønsted" acid. Which means that it is a compound capable of donating hydrogen ions to an acceptor molecule that functions as a Brønsted base..
Its synthesis was first achieved in 1838 and, since then, it is recognized as the "prototype" for a general class of molecular "rearrangements" for different synthetic purposes or for experimental research, including some "tracing" or "tracing" techniques. ”With isotopes.
Benzyl acid is used in the pharmaceutical industry for the synthesis of pharmaceutical glycolates, which are compounds that function as excipients for the transport and "delivery" of drugs such as clidinium, dilantin, mepenzolate, flutropium and others..
In addition, it has also been used successfully as an enhancer in pharmacological cosmetic products to treat acne, psoriasis, age spots, wrinkles and other common dermatological conditions..
Article index
- 1 Synthesis of benzyl acid
- 2 Transposition
- 3 Uses
- 3.1 Pharmacology
- 3.2 Other uses
- 4 References
Benzyl acid synthesis
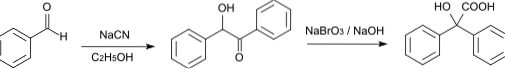
The synthesis of benzyl acid was first reported in 1838, when von Liebig described the rearrangement transformation of a common aromatic α-diketone (benzyl) into the salt of an α-hydroxy acid (benzyl acid), induced by an ion hydroxide.
This reaction of molecular "rearrangement" of benzyl to produce benzyl acid has to do with the migration of an aryl group (a substituent derived from an aromatic group, often an aromatic hydrocarbon), which is caused by the presence of a basic compound.
Since this process was probably one of the first known molecular rearrangement reactions, the benzyl-benzyl acid rearrangement mechanism has served as a model for many other processes in organic synthesis and in the field of organic physicochemistry..
At present, benzyl acid is commonly prepared from benzyl or benzoin, using potassium hydroxide as the "inducing" agent. During the course of the reaction, the intermediate compound known as "potassium benzylate" is formed, which is a stable carboxylate salt..
The acidification with hydrochloric acid of the medium where this reaction occurs is what subsequently promotes the formation of benzyl acid from potassium benzylate. Furthermore, many experimental reports also indicate that the reaction is carried out under high temperature conditions..
Transposition
Rearrangement (or benzyl-benzyl acid rearrangement) is the model reaction for various base-induced transformations of α-diketones into salts of α-hydroxy acids. This can be carried out with different types of diketones such as aromatic, semi aromatic, alicyclic, aliphatic or heterocyclic α-diketones, in order to produce benzyl acid..
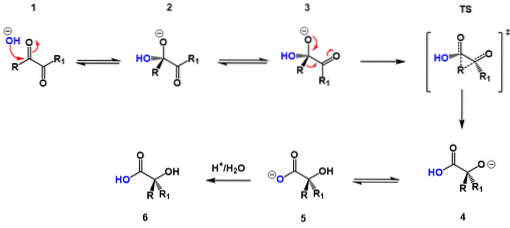
The reaction mechanism is basically the same in all cases. It consists of three steps:
1- The reversible addition of a hydroxide ion to a carbonyl group of the α-diketone compound, that is, to one of its ketone groups, which gives rise to a negatively charged intermediate that in many texts is known as a “hydroxyalkoxide”.
2- The intramolecular rearrangement of the intermediate, which produces a second intermediate that has a more suitable conformation for the nucleophilic attack that will take place on the carbonyl group.
3- The rapid conversion of the second intermediate into the salt of the corresponding α-hydroxy acid by means of a proton transfer reaction.
The α-diketones can also be transformed into benzyl acid esters through their reaction with an alcohol in the presence of an alkoxide ion..
The rearrangement, then, is nothing more than the organic reaction of an α-diketone in the presence of a base (usually potassium hydroxide) to produce the corresponding α-hydroxycarboxylic acids, as is the case in the synthesis of benzyl acid a starting from benzyl (1,2-diphenylethanedione).
As could be understood from this process, the rearrangement involves the intramolecular migration of a carbanion, which is an anionic organic compound where the negative charge resides on a carbon atom..
Applications
Benzyl acid, as discussed above, is an organic compound that serves as a "base" for the chemical synthesis of various types of compounds..
Pharmacology
Pharmaceutical glycolates are a good example of the practical benefits of benzyl acid. Glycolates are used as excipients, which are nothing more than substances that are mixed with drugs to give them flavor, shape, consistency or other properties (stability, for example), but which are inert from a pharmacological point of view.
Among the main drugs produced with excipients derived from benzyl acid are some compounds derived from:
- Clidinium: drug used to treat certain stomach disorders such as ulcers, irritable bowel syndrome, intestinal infections, abdominal cramps, among others. It works to reduce some body secretions and is usually achieved in the form of clidinium bromide..
- Dilantin: also known generically as phenytoin, which is an antiepileptic or anti-seizure drug, useful for some types of seizures.
- Mepenzolate: used to reduce heartburn and intestinal muscle spasms, as well as to treat stomach ulcers.
- Flutropium: anticholinergic agent that functions as a competitive antagonist of acetylcholine and is used mainly for the treatment of chronic obstructive pulmonary diseases.
In these drugs, the function of benzyl acid as an excipient is probably to transport the active compounds and, once inside the body, the bond between these can be hydrolyzed and the latter are released to exert their pharmacological functions..
Other uses
In addition to those already mentioned, benzyl acid is also used in pharmacological cosmetic formulations, where it has been proven effective for the treatment of acne, psoriasis, age spots, dry skin, pigmented skin lesions, wrinkles, etc..
References
- Drugs.com. (2018). Drugs.com: Know more. Be sure. Retrieved January 8, 2020, from www.drugs.com
- Drugs.com. (2019). Drugs.com: Know more. Be sure. Retrieved January 8, 2020, from www.drugs.com
- Drugs.ncats.io. National Center for Advancing Translational Sciences. Retrieved January 8, 2020, from www.drugs.ncats.io
- Everyday Health. Retrieved January 8, 2020, from www.everydayhealth.com
- Gill, G. B. (1991). Benzil-Benzilic Acid Rearrangements.
- National Center for Biotechnology Information. PubChem Database. Benzilic acid, CID = 6463, pubchem.ncbi.nlm.nih.gov
- Robertson, D. (2012). MiraCosta College. Retrieved January 8, 2020, from www.miracosta.edu
- Ruey, J. Y., & Van Scott, E. J. (1997). U.S. Patent No. 5,621,006. Washington, DC: U.S. Patent and Trademark Office.
- Selman, S., & Eastham, J. F. (1960). Benzilic acid and related rearrangements. Quarterly Reviews, Chemical Society, 14 (3), 221-235.
- Thornton, P. (2019). Drugs.com: Know more. Be sure. Retrieved January 8, 2020, from www.drugs.com



Yet No Comments