
Cinnamic acid structure, properties, production, uses

The cinnamic acid is an organic compound whose chemical formula is C6H5CHCHCOOH. It is a monocarboxylic acid. Its name comes from the plant Cinnamomum cassia, whose bark is aromatic like that of the cinnamon plant.
Other names are 3-phenyl-2-propenoic acid and β-phenyl acrylic acid. Many derivatives of cinnamic acid have been obtained, which exhibit therapeutic properties.

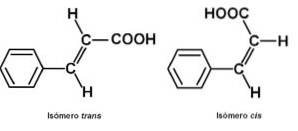
Due to the C = C double bond, the cinnamic acid molecule has two isomers: cis Y trans. The shape trans it is the most abundant in nature. Cinnamic acid trans It is a constituent of the essential oils of cinnamon, basil, black tea tree or Melaleuca bracteata, cocoa, tolu balsam, galangal major or Alpinia galanga and storach leaves. Cinnamic acid cis has been found in the plant of Alpinia malaccensis.
In the late 19th and early 20th centuries, cinnamic acid was studied and used to treat various diseases. However, its use was discontinued and research was resumed a few years ago, generating promising results..
Article index
- 1 Structure
- 2 Nomenclature
- 3 Properties
- 3.1 Physical state
- 3.2 Molecular weight
- 3.3 Melting point
- 3.4 Boiling point
- 3.5 Solubility
- 3.6 Chemical properties
- 3.7 Role in nature
- 4 Obtaining
- 5 Use in medical applications
- 5.1 Anti-cancer effect
- 5.2 Positive effects against insulin resistance and diabetes
- 5.3 Anti-tuberculosis effects
- 5.4 Various beneficial effects
- 6 Use in the food industry
- 7 New applications
- 8 References
Structure
The cinnamic acid molecule contains a phenyl group, a propenyl (propane with a C = C double bond) and a carboxyl group -COOH attached to the latter. It is like an acrylic acid with a phenyl group on the third carbon.
Due to its double bond, the molecule can be in the form cis or the way trans. In the way trans, groups C6H5- and -COOH are at diagonally opposite locations on the C = C double bond. In the way cis these groups are on the same side (one on each carbon of the C = C double bond).
Nomenclature
- Cinnamic acid
- 3-phenyl-2-propenoic acid
- Β-phenyl acrylic acid
- Benzenepropenoic acid
- Benzene acrylic acid
Properties
Physical state
White crystalline solid with monoclinic prism structure.

Molecular weight
148.16 g / mol
Melting point
Isomer cis: 68 ºC
Isomer trans: 133 ºC
Boiling point
Isomer cis: 125 ºC at 19 mm Hg
Isomer trans: 300 ºC
Solubility
Weakly soluble in water: 0.546 g / L at 25 ºC.
Very soluble in ether.
Chemical properties
The cinnamic acid molecule has three reactive sites: substitution in the benzene ring, addition in unsaturation (C = C double bond) and reaction of the carboxyl group -COOH.
This allows many derivatives to be obtained from cinnamic acid, which has generated special interest from researchers, especially in the field of medicine..
The presence of a benzene ring in its molecule and the unsaturated short chain make it have low polarity, hence its low solubility in water..
Cinnamic acid salts or alkali metal cinnamates are soluble in water.
Role in nature
Cinnamic acid has a particular role in the metabolism of plants, as it is distributed throughout the entire plant kingdom.

It is a precursor of flavonoids, alkaloids, coumarins and lignin, a structural component of plants.
It belongs to the class of auxins, which are hormones that regulate cell growth in plants.
Obtaining
In nature the isomer trans of cinnamic acid comes from the removal of ammonia from phenylalanine (an amino acid), which occurs thanks to the enzyme phenylalanine-ammonia-lyase. This enzyme is found in higher plants, fungi, and yeasts.
Cinnamic acid is often conjugated with amino acids and sugar acids. For example, cinnamoglycine is present in plants, but it could be a product of animal metabolism. It is found in the scent gland of the Canadian beaver, but you likely get it from food.
Use in medical applications
Anti-cancer effect
Cinnamic acid has been used for hundreds of years for the treatment of cancer in Chinese medicine, as it is part of the Xuanshen, which is the root of a herbaceous plant of the family of the Scrophulariaceae.
In the late 19th and early 20th centuries it was used to treat cancer, but research was not continued until a few decades ago.
Although its role is unknown, its effectiveness against bone cancer (osteosarcoma) has recently been confirmed.
It was also found to exert an antiproliferation effect on malignant cells of colon adenocarcinoma, melanoma, prostate and lung cancer..
According to some experiences, the treatment of melanoma cells with cinnamic acid for 3 days resulted in a 75-95% loss of invasiveness, that is, the ability to degrade and cross the tissue barrier. This is attributed to the modulation it exerts on the genes involved in tumor metastasis..
Furthermore, cinnamic acid has been found to be a potent inhibitor of an enzyme that influences the progression of hormone-dependent forms of cancer, such as prostate, breast, and endometrial cancer..
In addition, countless compounds derived from cinnamic acid have anticancer properties..
Positive effects against insulin resistance and diabetes
Some researchers found that cinnamic acid can alleviate insulin resistance.
This disease consists of the ineffectiveness of the action of insulin or the inability of it to achieve a normal response of glucose absorption by the cells.
Due to this, the cells do not absorb glucose, which remains in the blood and hyperglycemia or excess glucose in the blood occurs. The cell is then said to be insulin resistant.
The consequence of not treating insulin resistance is type 2 diabetes.
Cinnamic acid has mechanisms of action that improve the effectiveness of insulin, which translates into increased glucose uptake by insulin-resistant cells and disease relief..
Several polyphenolic derivatives of cinnamic acid are also beneficial in treating these diseases, but due to their low bioavailability, both cinnamic acid and its derivatives are still under study..
Efforts are being made to develop various forms of administration of these compounds, such as nanoparticles, encapsulation and emulsions..
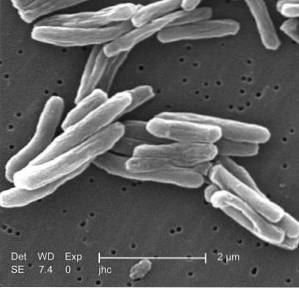
Anti-tuberculosis effects
Acid trans-Cinnamic and its derivatives were used as anti-tuberculosis agents around the year 1894. However, only recently have they been studied again for this purpose..
They have now been found to have a synergistic effect when used with other anti-tuberculosis drugs, even those towards which the bacteria Mycobacterium tuberculosis has become resistant, repowering the action of the drug.
Various beneficial effects
Cinnamic acid and many of its derivatives exhibit hepatoprotective, antimalarial, antioxidant and protective activity of the cardiovascular system.
Also many of its polyphenolic derivatives exert antibacterial, antiviral and antifungal effects.
Use in the food industry
Cinnamic acid and several of its derivatives are used to flavor some foods.
For cinnamic acid in particular the Daily Intake Limit, or ADI Acceptable Daily Intake), according to the standards of the Council of Europe (Council of Europe) is 1.25 mg / kg per day.
New applications
Recently (2019) it was found that acid cis-cinnamic favors the growth of the plant of Arabidopsis thaliana, which is a relative of cabbage and mustard.
The scientists concluded that the acid cis-Cinnamic has a high potential as an agrochemical that promotes plant growth..
References
- Hoskins, J.A. (1984). The Occurrence, Metabolism and Toxicity of Cinnamic Acid and Related Compounds. Journal of Applied Toxicology, Vol. 4, No. 6, 1984. Recovered from onlinelibrary.wiley.com.
- U.S. National Library of Medicine. (2019). Cinnamic acid. Recovered from: pubchem.ncbi.nlm.nih.gov.
- Huang, D.-W. et al. (2009). Effect of Caffeic Acid and Cinnamic Acid on Glucose Uptake in Insulin-Resistant Mouse Hepatocytes. Agric. Food Chem. 2009, 57, 7687-7692. Recovered from pubs.acs.org.
- De, P. et al. (2011). Cinnamic Acid Derivatives as Anticancer Agents - A Review. Current Medicinal Chemistry, 2011, 18, 1672-1703. Recovered from eurekaselect.com.
- De, P. et al. (2012). Cinnamic Acid Derivatives in Tuberculosis, Malaria and Cardiovascular Diseases - A Review. Current Organic Chemistry, 2012, 16, 747-768. Recovered from eurekaselect.com.
- Sova, M. (2012). Antioxidant and Antimicrobial Activities of Cinnamic Acid Derivatives. Mini-Reviews in Medicinal Chemistry, 2012, 12, 749-767. Recovered from ncbi.nlm.nih.org.
- Adisakwattana, S. (2017). Cinnamic Acid and Its Derivatives: Mechanisms for Prevention and Mangement of Diabetes and Its Complications. Nutrients 2017, 9, 163. Recovered from ncbi.nlm.nih.gov.
- Steenackers, W. et al. (2019). cis-Cinnamic acid is a natural plant growth-promoting compound. Exp. Bot. 2019 Aug 30. Recovered from ncbi.nlm.nih.gov.


Yet No Comments