
Polylactic acid structure, properties, synthesis, uses

The polylactic acid, whose correct name is poly- (lactic acid), it is a material formed by the polymerization of lactic acid. It is also known as poly-lactide, as it can be obtained from the breakdown and polymerization of lactide, which is a dimer of lactic acid..
Poly (lactic acid) or PLA is not an acid, it is a polyester, which can be observed in the monomer that makes it up. It is an easily biodegradable polymer and is biocompatible. Both properties are due to the fact that it can be easily hydrolyzed both in the environment and in the human or animal body. In addition, its degradation does not generate toxic compounds.
The involvement of PLA in suturing filaments during surgical operations has been known for years. It is also used in the pharmaceutical industry in slow-release drugs.
It is used in implants for the human body and there are a large number of studies for its use in biological tissues, as well as for three-dimensional (3D) printing for the most diverse applications.
Being one of the most biodegradable and non-toxic polymers, its producers have proposed the replacement of all petroleum-derived plastics that are currently used in thousands of applications with this material..
In addition, according to its manufacturers, as it comes from renewable sources, the production and use of PLA is a way to reduce the amount of CO.two that is generated when producing plastics from the petrochemical industry.
Article index
- 1 Structure
- 2 Nomenclature
- 3 Properties
- 3.1 Physical state
- 3.2 Molecular weight
- 3.3 Glass transition temperature
- 3.4 Melting point
- 3.5 Decomposition temperature
- 3.6 Density
- 4 Other properties
- 4.1 Mechanics
- 4.2 Biocompatibility
- 4.3 Biodegradability
- 4.4 Easy modification of its characteristics
- 5 Synthesis
- 6 Uses in medicine
- 6.1 Sutures
- 6.2 Pharmaceutical use
- 6.3 Implants
- 6.4 Biological tissue engineering
- 7 Use in textile materials
- 8 Various applications
- 9 Uses in engineering and agriculture
- 10 Recent Studies
- 11 References
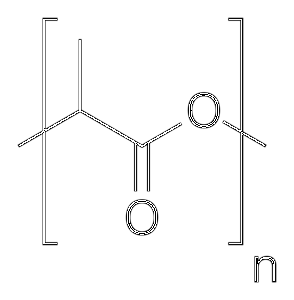
Structure
Poly- (lactic acid) is a polyester, that is, it has repeating ester units - (C = O) -O-R, something that can be seen in the following figure:
Nomenclature
- Poly- (lactic acid)
- Polylactide
- PLA
- Poly- (L-lactic acid) or PLLA
- Poly- (D, L-lactic acid) or PDLLA
- Polylactic acid
Properties
Physical state
- Poly (D, L-lactic acid): amorphous solid.
- Poly (L-lactic acid): brittle or brittle transparent semi-crystalline solid.
Molecular weight
It depends on the degree of polymerization of the material.
Glass transition temperature
It is the temperature below which the polymer is rigid, brittle and brittle, and above which the polymer becomes elastic and malleable.
- Poly (L-lactic acid): 63 ºC.
- Poly (D, L-lactic acid): 55 ºC.
Melting point
- Poly (L-lactic acid): 170-180 ºC.
- Poly (D, L-lactic acid): has no melting point because it is amorphous.
Decomposition temperature
227-255 ºC.
Density
- Amorphous: 1,248 g / cm3
- Crystalline: 1,290 g / cm3
Other properties
Mechanical
Poly- (L-lactic acid) has a higher mechanical strength than poly- (D, L-lactic acid).
PLA is easy to process thermoplastically, so very fine filaments can be obtained from this polymer..
Biocompatibility
Its degradation product, lactic acid, is non-toxic and totally biocompatible, because it is produced by living beings. In the case of humans, it is produced in the muscles and red blood cells.
Biodegradability
It can be thermally fractionated by hydrolysis in the human body, animals or microorganisms, which is called hydrolytic degradation.
Easy modification of its characteristics
Their physical, chemical and biological properties can be tailored through appropriate modifications, copolymerizations and grafts.
Synthesis
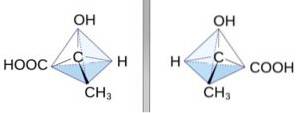
It was first obtained in 1932 by heating lactic acid under vacuum. HO-CH3-CH-COOH lactic acid is a molecule with a chiral center (that is, a carbon atom attached to four different groups).
For this reason it has two enantiomers or specular isomers (they are two molecules that are identical but with different spatial orientation of their atoms).
The enantiomers are L-lactic acid and D-lactic acid, which are distinguished from each other by the way they deflect polarized light. They are mirror images.
L-lactic acid is obtained from the fermentation by microorganisms of natural sugars such as molasses, potato starch or corn dextrose. This is the currently preferred way to obtain it.
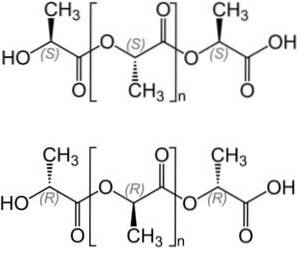
When poly (lactic acid) is prepared from L-lactic acid, you get poly (L-lactic acid), or PLLA.
On the other hand, when the polymer is prepared from a mixture of L-lactic acid and D-lactic acid, the poly- (D, L-lactic acid) or PDLLA is obtained.
In this case, the acid mixture is a combination in equal parts of the D and L enantiomers, obtained by synthesis from the ethylene of petroleum. This form of obtaining is rarely used today.
PLLA and PDLLA have slightly different properties. Polymerization can be carried out in two ways:
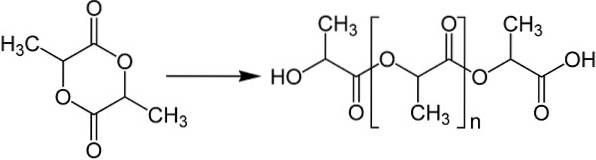
- Formation of an intermediate: the cyclic dimer called lactide, whose polymerization can be controlled and a product with the desired molecular weight can be obtained.
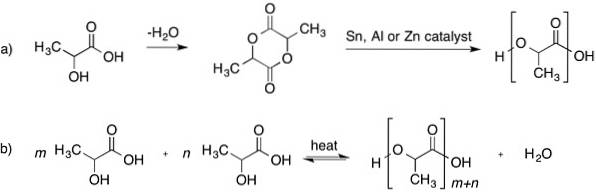
Uses in medicine
Its degradation products are non-toxic, which favors its application in this field..
Sutures
The basic requirement for suture filaments is that they hold tissues in place until natural healing provides strong tissue at the junction site..
Since 1972 a suture material called Vicryl has been manufactured, a very strong bioabsorbable filament or thread. This thread is made of a copolymer of glycolic acid and lactic acid (90:10), which at the suture site is rapidly hydrolyzed, so it is easily absorbed by the body.
It is estimated that in the human body, PLA degrades 63% in about 168 days and 100% in 1.5 years.
Pharmaceutical use
PLA's biodegradability makes it useful for the controlled release of medicinal products.
In most cases the drug is released gradually due to hydrolytic degradation and morphological changes of the reservoir (made with the polymer) that contains the medicinal product..
In other cases, the drug is released slowly through the polymer membrane..
Implants
PLA has proven effective in implants and supports for the human body. Good results have been obtained in the fixation of fractures and osteotomies or bone surgeries.
Biological tissue engineering
Currently, many studies are being carried out for the application of PLA in the reconstruction of tissues and organs..
PLA filaments have been developed for nerve regeneration in paralyzed patients.
The PLA fiber is previously treated by plasma to make it receptive to cell growth. The ends of the nerve to be repaired are joined by means of an artificial segment of PLA treated with plasma.
On this segment special cells are seeded which will grow and fill the void between the two ends of the nerve, joining them. Over time, the PLA support wears off leaving a continuous channel of nerve cells.
It has also been used in the reconstruction of bladders, acting as a scaffold or platform on which urothelial cells (cells that line the bladder and organs of the urinary system) and smooth muscle cells are seeded..
Use in textile materials
The chemistry of PLA allows the control of certain properties of the fiber that make it suitable for a wide variety of textile, clothing and furniture applications..
For example, its ability to absorb moisture, and at the same time low retention of moisture and odors, makes it useful for making clothing for high-performance athletes. It is hypoallergenic, does not irritate the skin.
It even works for pet clothes and does not require ironing. It has low density, so it is lighter than other fibers.
It comes from a renewable source and its production is economical.
Various applications
PLA is suitable for making bottles for various uses (shampoo, juices and water). These bottles have shine, transparency and clarity. In addition, PLA is an exceptional barrier to odors and flavors..
However, this use is for temperatures below 50-60 ºC, since it tends to deform when reaching those temperatures..
It is used in the production of disposable plates, cups and food utensils, as well as food containers, such as yogurt, fruits, pasta, cheese, etc., or PLA foam trays for packing fresh food. It does not absorb grease, oil, moisture and has flexibility. Compost can be made from waste PLA.

It is also used to make thin sheets to pack foods such as chips or other foods.

It can be used to make electronic transaction cards and hotel room key cards. PLA cards can meet security features and allow the application of magnetic tapes.
It is widely used to manufacture the boxes or covers of highly delicate products, such as electronic devices and cosmetics. Grades specially prepared for this use are used, by coupling with other fibers.
Expanded foam can be made from PLA to be used as a shock absorbing material for shipping delicate items or instruments.
Used to make toys for children.
Uses in engineering and agriculture
PLA is used to make drains in construction sites, floor construction materials such as carpets, laminate flooring and wall wallpaper, for carpets and car cushion fabrics..
Its use is under development in the electrical industry, as a coating for conductive wires..
Among its applications is that of agriculture, with PLA soil protective films are manufactured, which allow weed control and favor the retention of fertilizer. PLA films are biodegradable, they can be incorporated into the soil at the end of the harvest and thus provide nutrients.

Recent studies
The addition of nanocomposites to PLA is being studied to improve some of its properties, such as thermal resistance, crystallization speed, flame retardancy, antistatic and electrical conductive characteristics, anti-UV and antibacterial property..
Some researchers have managed to increase the mechanical strength and electrical conductivity of PLA by adding graphene nanoparticles. This considerably increases the applications that PLA can have in relation to 3D printing..
Other scientists succeeded in developing a vascular patch (to repair arteries in the human body) by grafting an organophosphate-phosphorylcholine onto a PLA scaffold or platform.
The vascular patch demonstrated such favorable properties as to be considered promising for vascular tissue engineering.
Its properties include the fact that it does not produce hemolysis (disintegration of red blood cells), it is not toxic to cells, resists the adhesion of platelets and has good affinity towards the cells that line the blood vessels..
References
- Mirae Kim, et al. (2019). Electrically Conducting and Mechanically Strong Graphene-Polylactic Acid Composites for 3D Printing. ACS Applied Materials & Interfaces. 2019, 11, 12, 11841-11848. Recovered from pubs.acs.org.
- Tin Sin, Lee et al. (2012). Applications of Poly (lactic Acid). In Handbook of Biopolymers and Biodegradable Plastics. Chapter 3. Recovered from sciencedirect.com.
- Gupta, Bhuvanesh, et al. (2007). Poly (lactic acid) fiber: An overview. Prog. Polym. Sci. 32 (2007) 455-482. Recovered from sciencedirect.com.
- Raquez, Jean-Marie et al. (2013). Polylactide (PLA) -based nanocomposites. Progress in Polymer Science. 38 (2013) 1504-1542. Recovered from sciencedirect.
- Zhang, Jun et al. (2019). Zwitterionic Polymer-Grafed Polylactic Acid Vascular Patches Based on Decellularized Scaffold for Tissue Engineering. ACS Biomaterials Science & Engineering. Publication Date: July 25, 2019. Recovered from pubs.acs.org.



Yet No Comments