
Sphingosine synthesis, structure, functions and diseases
The sphingosine It is a complex amino alcohol of great importance, because it is a precursor component of sphingolipids in general. The most relevant complex phospholipids or sphingolipids are sphingomyelin and glycosphingolipids. These fulfill specific functions in the preservation of the structure of the membranes of nerve cells, allowing them to fulfill their functions..
All sphingolipids have in common that they are formed with the same base substance, ceramide, which is made up of sphingosine plus Acetyl CoA, therefore it is also called N-acylphingosine.
Among the complex phospholipids, sphingomyelin is one of the most important and abundant in the brain and nervous tissue. It is mainly found as a constituent of the myelin sheath that covers the nerves.
While glycosphingolipids are glucose-containing sphingolipids. Among the most prominent are the cerebrosides (galactocerebroside and glucocerebroside) and gangliosides. The latter are involved in the transmission of nerve impulses, because they make up the nerve endings.
There are also others, such as globosides and sulphates, which are part of the plasma membranes of the entire organism, being important as membrane receptors..
Article index
- 1 Synthesis
- 2 Structure
- 3 Functions
- 3.1 Sphingosine
- 3.2 Derivative of sphingosine (sphingosine 1-phosphate)
- 4 Diseases caused by sphingosine deficiency
- 4.1 Farber's lipogranulomatosis or Farber's disease
- 5 References
Synthesis
The amino alcohol sphingosine is synthesized in the endoplasmic reticulum. The synthesis process occurs as follows:
The amino acid serine, once activated by binding to pyridoxal phosphate in the presence of manganese ions, binds to palmitoyl-CoA to form 3-ketosphinganine. This reaction releases COtwo.
Sphingosine is formed after two reducing stages. The first involves the enzyme 3-ketosphinganine reductase. This reaction uses NADPH as the donor of H+, forming dihydrosphingosine.
In the second stage, the enzyme sphinganine reductase acts, with the participation of a flavoprotein, where sphingosine is obtained.
On the other hand, sphingosine can be synthesized by sphingolipid catabolism. For example, when sphingomyelin is hydrolyzed, fatty acid, phosphoric acid, choline and sphingosine are generated..
Structure
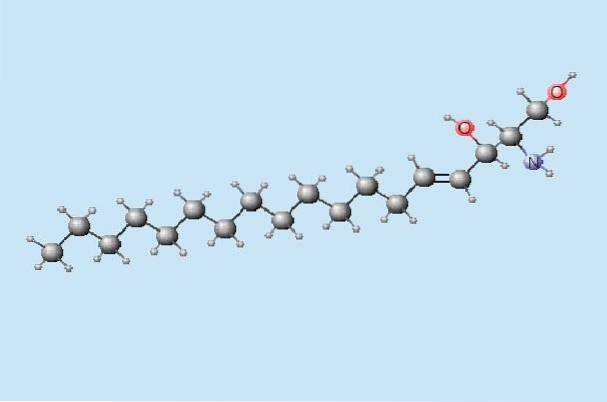
The chemical name for the amino alcohol sphingosine is 2-amino-4-octadecene-1,3-diol. The chemical structure can be described as a hydrocarbon chain composed of a total of 18 carbons, with an amino group and alcohol.
Features
Sphingosine
Under normal conditions, the sphingosine produced by the catabolism of sphingolipids is reused for the renewal and formation of new sphingolipids..
Sphingosine intervenes in cellular metabolic regulation processes related to lipid signaling pathways, as an extracellular mediator, acting on protein kinase C, which controls the enzymes involved during the cell growth and death process.
It also acts as an intracellular second messenger. This substance is capable of stopping the cell cycle, inducing the cell to programmed cell death or apoptosis..
Due to this function, it has generated interest in researchers as a therapy against cancer, together with tumor necrosis factor α.
An increase in the breakdown of sphingomyelins causes an accumulation of sphinganine and sphingosine (sphingoid bases). These substances in high concentrations inhibit the proper functioning of cell membranes.
This accumulation of sphingosine can occur in cases of poisoning due to consumption of grains contaminated with fumonisins, a type of mycotoxins produced by fungi of the genus Fusarium during storage..
Fumonisin inhibits the enzyme ceramide synthetase, which means that ceramide (N-acyl sphingosine) cannot be formed.
At the same time, it does not allow the synthesis of sphingomyelin, therefore sphingosine together with sphinganine are too concentrated, generating adverse effects..
Sphingosine derivative (sphingosine 1-phosphate)
From the phosphorylation of sphingosine by two enzymes (sphingosine kinase 1 and sphingosine kinase 2), its derivative called sphingosine 1-phosphate is formed..
Sphingosine 1-phosphate has the opposite effect to its precursor. It stimulates cell growth (mitogenic), preventing even the apoptotic action of some drugs used in cancer therapy, that is, its action is antiapoptotic..
This substance has been found in high concentrations in various malignant processes and tumor tissues. In addition, there is an exaggerated expression of the receptors of this lipid substance.
On the other hand, sphingosine 1-phosphate together with ceramide 1-phosphate acts in the regulation of immune cells, binding to the specific receptors present in said cells..
Lymphocytes especially present this type of receptors, being attracted by the presence of sphingosine 1-phosphate. In such a way that the lymphocytes leave the lymph nodes, pass to the lymph and later to the circulation.
Then they are concentrated in the place where the sphingolipid is being synthesized and this is how they participate in inflammatory processes.
Once lymphocytes bind to the substance through its receptor and induce a cellular response, they internalize the receptors, either to recycle them or to destroy them..
This action was observed by researchers, who have developed substances similar to sphingosine 1-phosphate to occupy specific receptors, in order to stimulate the internalization and destruction of the receptor, without causing cellular activation and thus reducing the immune response..
This type of substance is especially useful as immunosuppressive therapy in autoimmune diseases such as multiple sclerosis..
Diseases caused by sphingosine deficiency
Farber's lipogranulomatosis or Farber's disease
This is a rare disease with an autosomal recessive hereditary nature, very rare, with only 80 cases reported worldwide..
The cause of the disease is a mutation in the ASAH1 gene that codes for the lysosomal enzyme acid ceramidase. This enzyme has the function of hydrolyzing the ceramide and converting it into sphingosine and fatty acids.
The lack of the enzyme causes an accumulation of ceramide, a deficiency that manifests itself in the first months of life (3 - 6 months). The disease does not manifest itself in the same way in all affected individuals, with mild, moderate and severe cases being observed..
Mild cases have a longer life expectancy, and can reach adolescence and even adulthood, but the severe form is always fatal at the beginning of life.
Among the most frequent clinical manifestations of the disease are: severe hoarseness due to involvement in the larynx that can lead to aphonia due to inflammation of the vocal cords, dermatitis, skeletal deformations, pain, inflammation, paralysis, neurological deterioration or mental retardation.
In severe cases, it can present with hydrops fetalis, hepatosplenomegaly, lethargy and granulomatous infiltrations in the lungs and organs of the reticuloendothelial system such as the spleen and liver, with a very short life expectancy.
For cases with a longer life expectancy, there is no specific treatment, only the symptoms are treated..
References
- Torres-Sánchez L, López-Carrillo L. Consumption of fumonisins and damage to human health. Public health Mex. 2010; 52 (5): 461-467. Available at: scielo.org.
- Baumruker T, Bornancin F, Billich A. The role of sphingosine and ceramide kinases in inflammatory responses. Immunol Lett.2005; 96 (2): 175-85.
- Ponnusamy S, Meyers-Needham M, Senkal CE, et al. Sphingolipids and cancer: ceramide and sphingosine-1-phosphate in the regulation of cell death and drug resistance. Future Oncol. 2010; 6 (10): 1603-24.
- Bazua-Valenti S; Garcia-Sainz A. Sphingosine 1-phosphate and its S1P1 receptor: regulators of the immune response. Fac. Med. (Mex.), 2012; 55 (6): 53-57. Available in Scielo. Org
- Murray R, Granner D, Mayes P, Rodwell V. (1992). Harper's Biochemistry. 12 ava edition, Editorial El Manual Moderno. DF Mexico.


Yet No Comments