
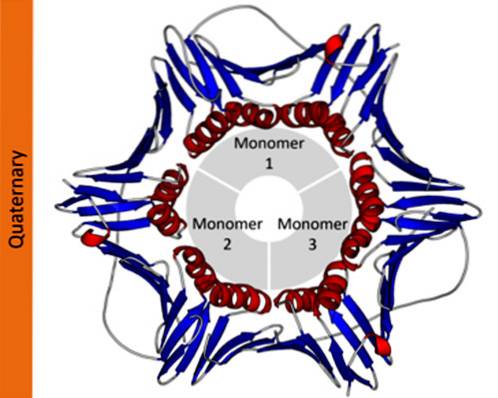
Quaternary structure of characteristic proteins
The quaternary structure of proteins defines the spatial relationships between each of its polypeptide subunits joined by non-covalent forces. In polymeric proteins, each of the polypeptide chains that make it up are called subunits or protomers.
Proteins can be made up of one (monomeric), two (dimeric), several (oligomeric), or many protomers (polymeric). These protomers can have a similar or very different molecular structure. In the first case they are said to be homotypic proteins and in the second case, heterotypic.
In scientific notation, biochemists use subscript Greek letters to describe the protomer composition of proteins. For example, a tetrameric homotypic protein is designated as α4, while a tetrameric protein made up of two different dimers is designated as αtwoβtwo.
Article index
- 1 Protein structure
- 1.1 Primary structure
- 1.2 Secondary structure
- 1.3 Tertiary structure
- 1.4 Quaternary structure
- 2 Stability of the quaternary structure
- 2.1 Hydrophobic interactions
- 2.2 Van der Waals interactions
- 2.3 Load-load interactions
- 2.4 Hydrogen bonds
- 2.5 Interactions of dipoles
- 3 Interactions between protomers
- 3.1 Homotypic interactions
- 3.2 Heterotypic interactions
- 4 References
Protein structure
Proteins are complex molecules that take on different three-dimensional configurations. These configurations are unique to each protein and allow them to perform very specific functions. The levels of structural organization of proteins are as follows.
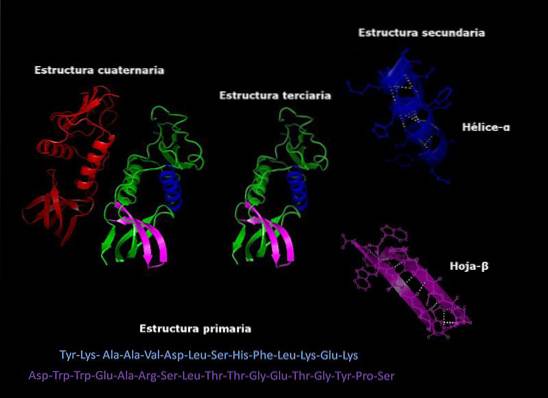
Primary structure
It refers to the sequence in which the different amino acids are arranged in the polypeptide chain. This sequence is given by the DNA sequence that encodes said protein.
Secondary structure
Most proteins are not fully extended long chains of amino acids, but rather have regions that are regularly folded into helixes or sheets. This folding is the one that receives the name of secondary structure.
Tertiary structure
The folded areas of the secondary structure can, in turn, be folded and assembled into more compact structures. This last fold is what gives the protein its three-dimensional shape..
Quaternary structure
In proteins that are made up of more than one subunit, the quaternary structures are the spatial relationships that exist between each subunit, which are linked by non-covalent bonds..
Quaternary structure stability
The three-dimensional structure of proteins is stabilized by weak or non-covalent interactions. While these bonds or interactions are much weaker than normal covalent bonds, they are numerous and their cumulative effect is powerful. Here we will see some of the most common interactions.
Hydrophobic interactions
Some amino acids contain hydrophobic side chains. When proteins have these amino acids, the folding of the molecule orders these side chains towards the interior of the protein and protects them from water. The nature of the different side chains means that they contribute in different ways to the hydrophobic effect..
Van der Waals interactions
These interactions occur when molecules or atoms that are not linked by covalent bonds get too close to each other, and because of this their outermost electronic orbitals begin to overlap..
At that moment a repulsive force is established between these atoms that grows very rapidly as their respective centers approach. These are the so-called "van der Waals forces".
Load-load interactions
It is the electrostatic interaction that occurs between a pair of charged particles. In proteins, this type of interaction occurs, both due to the net electrical charge of the protein and the individual charge of the ions contained within it. This type of interaction is sometimes called a salt bridge..
Hydrogen bonds
A hydrogen bond is established between a hydrogen atom covalently bonded to a hydrogen bond donor group and a pair of free electrons belonging to a bond acceptor group.
This type of bond is very important, since the properties of many molecules, including those of water and biological molecules, are largely due to hydrogen bonds. It shares properties of covalent bonds (electrons are shared) and also of non-covalent interactions (charge-charge interaction).
Dipole interactions
In molecules, including proteins, that do not have a net charge, a non-uniform arrangement of their internal charges can occur, with one extreme slightly more negative than the other. This is what is known as a dipole.
This dipolar condition of the molecule can be permanent, but it can also be induced. Dipoles can be attracted to ions or other dipoles. If the dipoles are permanent, the interaction has a greater range than that which occurs with induced dipoles.
In addition to these non-covalent interactions, some oligomeric proteins stabilize their quaternary structure through a type of covalent bond, the disulfide bond. These are established between the sulfhydryl groups of the cysteines of different protomers.
Disulfide bonds also help stabilize the secondary structure of proteins, but in this case, they link cysteinyl residues within the same polypeptide (intrapolypeptide disulfide bonds)..
Interactions between protomers
As noted above, in proteins that are made up of several subunits or protomers, these subunits can be similar (homotypic) or different (heterotypic)..
Homotypic interactions
The subunits that make up a protein are asymmetric polypeptide chains. However, in homotypic interactions, these subunits can associate in different ways, achieving different types of symmetry..
The interacting groups of each protomer are generally located in different positions, which is why they are called heterologous interactions. Heterologous interactions between the different subunits sometimes occur in such a way that each subunit is twisted with respect to the preceding one, being able to achieve a helical structure.
On other occasions the interactions occur in such a way that defined groups of subunits are arranged around one or more axes of symmetry, in what is known as point-group symmetry. When there are several axes of symmetry, each subunit rotates with respect to its neighbor 360 ° / n (where n represents the number of axes).
Among the types of symmetry that are obtained in this way are, for example, helical, cubic and icosahedral.
When two subunits interact through a binary axis, each unit rotates 180 ° with respect to the other, around that axis. This symmetry is known as C symmetrytwo. In it, the interaction sites in each subunit are identical; in this case we do not speak of a heterologous interaction, but an isologous interaction.
If, on the contrary, the association between the two components of the dimer is heterologous, then an asymmetric dimer will be obtained.
Heterotypic interactions
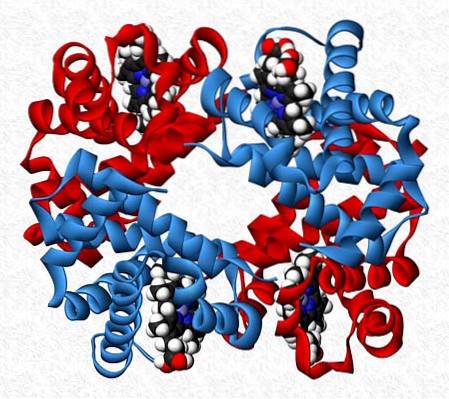
The subunits that interact in a protein are not always of the same nature. There are proteins that are made up of twelve or more different subunits.
The interactions that maintain the stability of the protein are the same as in homotypic interactions, but completely asymmetric molecules are generally obtained..
Hemoglobin, for example, is a tetramer that has two pairs of different subunits (αtwoβtwo).
References
- C.K. Mathews, K.E. van Holde & K.G. Ahern (2002). Biochemestry. 3rd edition. Benjamin / Cummings Publishing Company, Inc.
- R.K. Murray, P. Mayes, D.C. Granner & V.W. Rodwell (1996). Harper's Biochemestry. Appleton & Lange
- J.M. Berg, J.L. Tymoczko & L. Stryer (2002). Biochemestry. 5th edition. W. H. Freeman and Company.
- J. Koolman & K.-H. Roehm (2005). Color Atlas of Biochemistry. 2nd edition. Thieme.
- A. Lehninger (1978). Biochemistry. Ediciones Omega, S.A.
- L. Stryer (1995). Biochemestry. W.H. Freeman and Company, New York.



Yet No Comments