
Phosphatidylethanolamine structure, biosynthesis and functions
The phosphatidylethanolamine (PE) is a glycerophospholipid abundant in the plasma membranes of prokaryotic organisms. On the contrary, in eukaryotic cell membranes this is the second most abundant glycerophospholipid on the inside of the plasma membrane after phosphatidylcholine..
Despite the abundance of phosphatidylethanolamine, its abundance depends not only on the cell type but also on the compartment and the moment of the specific cell life cycle considered..
Biological membranes are barriers that define cellular organisms. Not only do they have protection and isolation functions, but they are also key to the establishment of proteins that require a hydrophobic environment for their optimal functioning..
Both eukaryotes and prokaryotes have membranes composed mainly of glycerophospholipids and to a lesser extent sphingolipids and sterols..
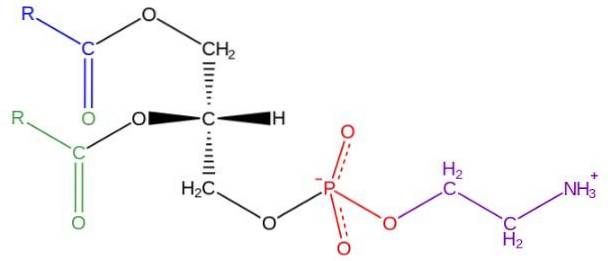
Glycerophospholipids are amphipathic molecules structured on an L-glycerol backbone that is esterified at the sn-1 and sn-2 positions by two fatty acids of varying length and degree of saturation. In the hydroxyl of the sn-3 position it is esterified by a phosphate group, to which in turn different types of molecules can be attached that give rise to the various classes of glycerophospholipids.
In the cellular world there is a great variety of glycerophospholipids, however, the most abundant are phosphatidylcholine (PC), phosphatidylethanolamine (PE), phosphatidylserine (PS), phosphatidylinositol (PI), phosphatidic acid (PA), phosphatidylglycerol (PG) and cardiolipin (CL).
Article index
- 1 Structure
- 2 Biosynthesis
- 2.1 Kennedy Route
- 2.2 PSD path
- 3 Functions
- 4 References
Structure
The structure of phosphatidylethanolamine was discovered by Baer et al. In 1952. As has been determined experimentally for all glycerophospholipids, phosphatidylethanolamine is formed by a glycerol molecule esterified at the sn-1 and sn-2 positions with acid chains fatty of between 16 and 20 carbon atoms.
The fatty acids esterified in the sn-1 hydroxyl are generally saturated (without double bonds) with maximum lengths of 18 carbon atoms, while the chains linked in the sn-2 position are longer and with one or more unsaturations ( double bonds).
The degree of saturation of these chains contributes to the elasticity of the membrane, which has a great influence on the insertion and sequestration of proteins in the bilayer..
Phosphatidylethanolamine is considered a non-lamellar glycerophospholipid, since it has a conical geometric shape. This shape is given by the small size of its polar group or "head", in relation to that of the fatty acid chains that comprise the hydrophobic "tails".
The "head" or polar group of phosphatidylethanolamine has a zwitterionic character, that is, it has groups that can be positively and negatively charged under certain pH conditions..
This characteristic allows it to hydrogen bond with a large number of amino acid residues and its charge distribution is an essential determinant for the domain topology of many integral membrane proteins..
Biosynthesis
In eukaryotic cells the synthesis of structural lipids is geographically restricted, the main biosynthesis site being the endoplasmic reticulum (ER) and to a lesser extent the Golgi apparatus..
There are four independent biosynthetic pathways for the production of phosphatidylethanolamine: (1) the CDP-ethanolamine pathway, also known as the Kennedy pathway; (2) the PSD pathway for phosphatidylserine (PS) decarboxylation; (3) acylation of lyso-PE and (4) base change reactions of the polar group of other glycerophospholipids.
Kennedy Route
The biosynthesis of phosphatidylethanolamine by this route is limited to the ER and it has been shown that in hamster liver cells it is the main route of production. It consists of three consecutive enzymatic steps catalyzed by three different enzymes.
In the first step, phosphoethanolamine and ADP are produced thanks to the action of ethanolamine kinase, which catalyzes the ATP-dependent phosphorylation of ethanolamine..
Unlike plants, neither mammals nor yeasts are capable of producing this substrate, so it must be consumed in the diet or obtained from the degradation of pre-existing phosphatidylethanolamine or sphingosine molecules..
Phosphoethanolamine is used by CTP: phosphoethanolamine cytidyltransferase (ET) to form the high-energy compound CDP: ethanolamine and an inorganic phosphate.
1,2-Diacylglycerol ethanolamine phosphotransferase (ETP) uses the energy contained in the CDP-ethanolamine bond to covalently bind ethanolamine to a membrane-inserted diacylglycerol molecule, giving rise to phosphatidylethanolamine..
Route PSD
This route operates both in prokaryotes and in yeast and mammals. In bacteria it occurs in the plasma membrane, but in eukaryotes it occurs in an area of the endoplasmic reticulum that is closely related to the mitochondrial membrane.
In mammals the pathway is catalyzed by a single enzyme, phosphatidylserine decarboxylase (PSD1p), which is embedded in the mitochondrial membrane, whose gene is encoded by the nucleus. The reaction involves decarboxylation of PS to phosphatidylethanolamine.
The remaining two pathways (PE-lyso acylation and polar group-dependent calcium exchange) occur in the endoplasmic reticulum, but do not contribute significantly to total phosphatidylethanolamine production in eukaryotic cells..
Features
Glycerophospholipids have three main functions in the cell, among which the structural functions, energy storage and cell signaling stand out..
Phosphatidylethanolamine is associated with the anchoring, stabilization and folding of multiple membrane proteins, as well as the conformational changes necessary for the functioning of many enzymes.
There is experimental evidence that proposes phosphatidylethanolamine as a crucial glycerophospholipid in the late stage of telophase, during the formation of the contractile ring and the establishment of the fragmoplast that allows the division of the membrane of the two daughter cells..
It also has an important function in all the processes of fusion and fission (union and separation) of the membranes of both the endoplasmic reticulum and the Golgi apparatus..
In E. coli it has been shown that phosphatidylethanolamine is necessary for the correct folding and function of the enzyme lactose permease, which is why it has been suggested that it plays a role as a molecular “chaperone”.
Phosphatidylethanolamine is the main donor of the ethanolamine molecule necessary for the post-translational modification of numerous proteins, such as GPI anchors..
This glycerophospholipid is the precursor of numerous molecules with enzymatic activity. In addition, molecules derived from its metabolism, as well as diacylglycerol, phosphatidic acid and some fatty acids, can act as second messengers. Additionally, it is an important substrate for the production of phosphatidylcholine..
References
- Brouwers, J. F. H. M., Vernooij, E. A. A. M., Tielens, A. G. M., & van Golde, L. M. G. (1999). Rapid separation and identification of phosphatidylethanolamine molecular species. Journal of Lipid Research, 40 (1), 164-169. Recovered from jlr.org
- Calzada, E., McCaffery, J. M., & Claypool, S. M. (2018). Phosphatidylethanolamine produced in the inner mitochondrial membrane is essential for yeast cytochrome bc1 complex function 3. BioRxiv, 1, 46.
- Calzada, E., Onguka, O., & Claypool, S. M. (2016). Phosphatidylethanolamine Metabolism in Health and Disease. International Review of Cell and Molecular Biology (Vol. 321). Elsevier Inc.
- Gibellini, F., & Smith, T. K. (2010). The Kennedy pathway-de novo synthesis of phosphatidylethanolamine and phosphatidylcholine. IUBMB Life, 62 (6), 414-428.
- Harayama, T., & Riezman, H. (2018). Understanding the diversity of membrane lipid composition. Nature Reviews Molecular Cell Biology, 19 (5), 281-296.
- Luckey, M. (2008). Membrane structural biology: with biochemical and biophysical foundations. Cambrudge University Press. Recovered from cambrudge.org
- Seddon, J. M., Cevc, G., Kaye, R. D., & Marsh, D. (1984). X-ray Diffraction Study of the Polymorphism of Hydrated Diacyl- and Dialkylphosphatidylethanolamines. Biochemistry, 23 (12), 2634-2644.
- Sendecki, A. M., Poyton, M. F., Baxter, A. J., Yang, T., & Cremer, P. S. (2017). Supported Lipid Bilayers with Phosphatidylethanolamine as the Major Component. Langmuir, 33 (46), 13423-13429.
- van Meer, G., Voelker, D. R., & Feignenson, G. W. (2008). Membrane lipids: where they are and how they behave. Nature Reviews, 9, 112-124.
- Vance, J. E. (2003). Molecular and Cell Biology of Phosphatidylserine and Phosphatidylethanolamine Metabolism. In K. Moldave (Ed.), Progress Nucleic Acid Research and Molecular Biology (pp. 69-111). Academic Press.
- Vance, J. E. (2008). Phosphatidylserine and phosphatidylethanolamine in mammalian cells: two metabolically related aminophospholipids. Journal of Lipid Research, 49 (7), 1377-1387.
- Vance, J. E., & Tasseva, G. (2013). Formation and function of phosphatidylserine and phosphatidylethanolamine in mammalian cells. Biochimica et Biophysica Acta - Molecular and Cell Biology of Lipids, 1831 (3), 543-554.
- Watkins, S. M., Zhu, X., & Zeisel, S. H. (2003). Phosphatidylethanolamine-N-methyltransferase activity and dietary choline regulate liver-plasma lipid flux and essential fatty acid metabolism in mice. The Journal of Nutrition, 133 (11), 3386-3391.


Yet No Comments