
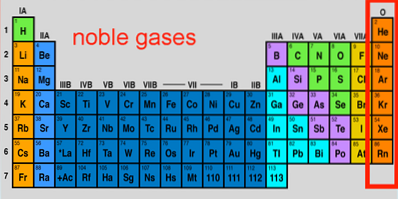
Noble gases characteristics, configuration, reactions, uses

The Noble gases They are a set of elements that are found in group 18 of the periodic table. Over the years they have also been called rare or inert gases, both of which are inaccurate; some of them are very abundant outside and inside planet Earth, and they are also capable, under extreme conditions, of reacting.
Its seven elements make up perhaps the most unique group in the periodic table, whose properties and low reactivities are as impressive as that of the noble metals. Among them parade the most inert element (neon), the second most abundant of the Cosmos (helium), and the heaviest and most unstable (oganeson).

Noble gases are the coldest substances in nature; withstand very low temperatures before condensing. Even more difficult is its freezing, since its intermolecular forces based on the London scattering, and the polarizability of its atoms, are too weak to barely keep them cohesive in a crystal..
Due to their low reactivity, they are relatively safe gases to store and do not pose too many risks. However, they can displace oxygen from the lungs and cause suffocation if inhaled excessively. On the other hand, two of its members are highly radioactive elements and, therefore, fatal to health..
The low reactivity of noble gases is also used to provide the reactions with an inert atmosphere; so that no reagent or product runs the risk of oxidizing and affecting the performance of the synthesis. This also favors electric arc welding processes..
On the other hand, in their liquid states they are excellent cryogenic refrigerants that guarantee the lowest temperatures, essential for the correct operation of highly energetic equipment, or for some materials to reach states of superconductivity.
Article index
- 1 Characteristics of noble gases
- 2 The 7 noble gases
- 3 Electronic configuration
- 4 Polarizability
- 5 Reactions
- 5.1 Helium and neon
- 5.2 Argon and krypton
- 5.3 Xenon and radon
- 6 Production
- 6.1 Air liquefaction
- 6.2 Distillation of natural gas and radioactive minerals
- 7 Dangers
- 8 Uses
- 8.1 Industry
- 8.2 Balloons and breathing tanks
- 8.3 Medicine
- 8.4 Others
- 9 References
Noble gas characteristics
Perhaps noble gases are the elements that share the most qualities in common, both physical and chemical. Its main characteristics are:
- All of them are colorless, odorless and tasteless; but when they are enclosed in ampoules at low pressures, and they receive an electric shock, they ionize and give off colorful lights (top image).
- Each noble gas has its own light and spectrum.
- They are monatomic species, the only ones in the periodic table that can exist in their respective physical states without the participation of chemical bonds (since metals are joined by metallic bonding). Therefore, they are perfect for studying the properties of gases, as they adapt very well to the spherical model of an ideal gas..
- These are generally the elements with the lowest melting and boiling points; so much so, that helium cannot even crystallize at absolute zero without an increase in pressure.
- Of all the elements they are the least reactive, even less so than the noble metals.
- Their ionization energies are the highest, as well as their electronegativities, assuming they form purely covalent bonds..
- Its atomic radii are also the smallest because they are at the right ends of each period..
The 7 noble gases
The seven noble gases are, from top to bottom, descending through group 18 of the periodic table:
-Helium, He
-Neon, Ne

-Argon, Ar
-Krypton, Kr
-Xenon, Xe

-Radon, Rn
-Oganeson, Og
All of them, except for the unstable and artificial oganeson, have been studied for their physical and chemical properties. Oganeson, due to its large atomic mass, is believed to not even be a gas, but rather a noble liquid or solid. Little is known about radon, because of its radioactivity, in relation to helium or argon.
Electronic configuration
Noble gases have been said to have their valence shell fully filled. So much so, that their electronic configurations are used to simplify that of other elements by using their symbols enclosed in square brackets ([He], [Ne], [Ar], etc.). Its electronic configurations are:
-Helium: 1stwo, [He] (2 electrons)
-Neon: 1stwo2stwo2 P6, [Ne] (10 electrons)
-Argon: 1stwo2stwo2 P63stwo3p6, [Ar] (18 electrons)
-Krypton: 1stwo2stwo2 P63stwo3p63d104stwo4p6, [Kr] (36 electrons)
-Xenon: 1stwo2stwo2 P63stwo3p63d104stwo4p64d105stwo5 p6, [Xe] (54 electrons)
-Radon: 1stwo2stwo2 P63stwo3p63d104stwo4p64d104f145stwo5 p65 d106stwo6p6, [Rn] (86 electrons)
The important thing is not to remember them, but to detail that they end in nstwonp6: the valence octet. Likewise, it is appreciated that its atoms have many electrons, which due to the great effective nuclear force are in a smaller volume compared to that of the other elements; that is, their atomic radii are smaller.
Therefore, their electronically dense atomic radii exhibit a chemical characteristic that all noble gases share: they are difficult to polarize..
Polarizability
Noble gases can be imagined as spheres of electron clouds. While descending through group 18, its radii increase, and in the same way the distance that separates the nucleus from the valence electrons (those of the nstwonp6).
These electrons feel a less attractive force on the part of the nucleus, they can move more freely; the spheres are more easily deformed the more voluminous they are. As a consequence of such movements, regions of low and high electron densities appear: the δ + and δ poles-.
When the atom of a noble gas is polarized, it becomes an instantaneous dipole capable of inducing another to the neighboring atom; that is, we are facing the dispersive forces of London.
That is why intermolecular forces increase from helium to radon, reflected in their increasing boiling points; and not only that, but their reactivities are also increased.
As atoms become more polarized, there is a greater possibility that their valence electrons participate in chemical reactions, after which noble gas compounds are generated..
Reactions
Helium and neon
Among the noble gases, the least reactive are helium and neon. In fact, neon is the most inert element of all, even though its electronegativity (from forming covalent bonds) exceeds that of fluorine.
None of its compounds are known under terrestrial conditions; However, in the Cosmos the existence of the molecular ion HeH is quite probable+. Likewise, when they are electronically excited they are capable of interacting with gaseous atoms and forming short-lived neutral molecules called excimers; such as HeNe, CsNe and Netwo.
On the other hand, although they are not considered compounds in a formal sense, He and Ne atoms can give rise to Van der Walls molecules; that is, compounds that are held "together" simply by dispersive forces. For example: Ag3He, HeCO, HeItwo, CF4Ne, Ne3Cltwo and NeBeCO3.
Likewise, such Van der Walls molecules can exist thanks to weak ion-induced dipole interactions; for example: Na+I have8, Rb+He, Cu+Ne3 and Cu+Ne12. Note that it is even possible for these molecules to become agglomerates of atoms: clusters.
And finally, He and Ne atoms can be "trapped" or intercalated in endohedral complexes of fullerenes or clathrates, without reacting; for example: [email protected]60, (Ntwo)6Ne7, He (HtwoOR)6 and Ne • NH4Fe (HCOO)3.
Argon and krypton
The noble gases argon and krypton, because they are more polarizable, tend to present more "compounds" than helium and neon. However, a part of them are more stable and characterizable, as they have a longer life span. Among some of them is HArF, and the molecular ion ArH+, present in nebulae by the action of cosmic rays.
From krypton begins the possibility of obtaining compounds in extreme, but sustainable conditions. This gas reacts with fluorine according to the following chemical equation:
Kr + Ftwo → KrFtwo
Note that krypton acquires an oxidation number of +2 (Krtwo+) thanks to fluoride. The KrFtwo in fact it can be synthesized in marketable amounts as an oxidizing and fluorinating agent.
Argon and krypton can establish a wide repertoire of clathrates, endohedral complexes, Van der Walls molecules, and some compounds awaiting discovery after their predicted existence..
Xenon and radon
Xenon is, among the noble gases, the king of reactivity. It forms the truly stable, marketable and characterizable compounds. In fact, its reactivity resembles that of oxygen under the appropriate conditions..
His first synthesized compound was “XePtF6", In 1962 by Neil Bartlett. This salt actually, according to the literature, consisted of a complex mixture of other fluorinated salts of xenon and platinum.
However, this was more than enough to demonstrate the affinity between xenon and fluorine. Among some of these compounds we have: XeFtwo, XeF4, XeF6 and [XeF]+[PtF5]-. When the XeF6 dissolves in water, generates an oxide:
XeF6 + 3 HtwoO → XeO3 + 6 HF
This XeO3 can originate the species known as xenatos (HXeO4-) or xenic acid (HtwoXeO4). Xenates disproportionate to perxenates (XeO64-); and if the medium is then acidified, in peroxenic acid (H4XeO6), which is dehydrated to xenon tetroxide (XeO4):
H4XeO6 → 2 HtwoO + XeO4
Radon should be the most reactive of the noble gases; But it is so radioactive that it hardly has time to react before disintegrating. The only compounds that have been fully synthesized are its fluoride (RnFtwo) and oxide (RnO3).
Production
Air liquefaction
The noble gases become more abundant in the Universe as we descend through group 18. In the atmosphere, however, helium is scarce, since the gravitational field of the Earth cannot retain it unlike other gases. That is why it was not detected in the air but in the Sun.
On the other hand, in the air there are notable amounts of argon, coming from the radioactive decay of the radioisotope 40K. Air is the most important natural source of argon, neon, krypton and xenon on the planet.
To produce them, the air must first be liquefied so that it condenses into a liquid. Then, this liquid undergoes a fractional distillation, thus separating each of the components of its mixture (Ntwo, ORtwo, COtwo, Ar, etc.).
Depending on how low the temperature and the abundance of the gas must be, its prices increase, ranking xenon as the most expensive, while helium as the cheapest..
Distillation of natural gas and radioactive minerals
Helium, for its part, is obtained from another fractional distillation; but not from the air, but from natural gas, enriched with helium thanks to the release of alpha particles from the radioactive minerals thorium and uranium.
Likewise, radon is "born" from the radioactive decay of radium in its respective minerals; but due to their lower abundance, and the short half-life of the Rn atoms, their abundance is laughable compared to that of their congeners (the other noble gases).
And finally, oganeson is a highly radioactive, ultramassic, man-made noble “gas” that can only exist briefly under controlled conditions within a laboratory..
Dangers
The main risk of noble gases is that they limit the use of oxygen by man, especially when an atmosphere with a high concentration of them is produced. That is why it is not recommended to inhale them excessively..
In the United States, a high concentration of radon has been detected in soils rich in uranium, which due to its radioactive characteristics could be a health risk.
Applications
Industry
Helium and argon are used to create an inert atmosphere for protection during welding and cutting. In addition, they are used in the manufacture of silicon semiconductors. Helium is used as a fill gas in thermometers.
Argon, in combination with nitrogen, is used in the manufacture of incandescent lamps. Krypton mixed with halogens, such as bromine and iodine, is used in discharge lamps. Neon is used in light signs, mixed with phosphors and other gases to tint its red color.
Xenon is used in arc lamps that emit light that resembles daylight, which are used in car headlights and projectors. The noble gases are mixed with halogens to produce ArF, KrF or XeCl, which are used in the production of excimer lasers.
This type of laser produces short-wave ultraviolet light that produces high-precision images and is used in the manufacture of integrated circuits. Helium and neon are used as cryogenic refrigerant gases.
Balloons and breathing tanks
Helium is used as a substitute for nitrogen in the respiratory gas mixture, due to its low solubility in the body. This avoids the formation of bubbles during the decompression phase during the ascent, in addition to eliminating the nitrogen narcosis.
Helium has replaced hydrogen as the gas that allows the elevation of airships and hot air balloons, because it is a light and non-combustible gas..
Medicine
Helium is used in the manufacture of superconducting magnets used in nuclear magnetic resonance equipment: a multi-application tool in medicine.
Krypton is used in halogen lamps used in laser eye surgery and angioplasty. Helium is used to facilitate breathing in asthmatic patients.
Xenon is used as an anesthetic due to its high lipid solubility, and is thought to be the anesthetic of the future. Xenon is also used in lung medical imaging.
Radon, a radioactive noble gas, is used in radiation therapy for some types of cancer.
Others
Argon is used in the synthesis of compounds substituting nitrogen as an inert atmosphere. Helium is used as a carrier gas in gas chromatography, as well as in Geiger counters to measure radiation.
References
- Shiver & Atkins. (2008). Inorganic chemistry. (Fourth edition). Mc Graw Hill.
- Whitten, Davis, Peck & Stanley. (2008). Chemistry. (8th ed.). CENGAGE Learning.
- Helmenstine, Anne Marie, Ph.D. (June 06, 2019). Noble Gases Properties, Uses and Sources. Recovered from: thoughtco.com
- Wikipedia. (2019). Noble gas. Recovered from: en.wikipedia.org
- Philip Ball. (January 18, 2012). Impossible chemistry: Forcing noble gases to work. Recovered from: newscientist.com
- Professor Patricia Shapley. (2011). Noble Gas Chemistry. Recovered from: butane.chem.uiuc.edu
- Gary J. Schrobilgen. (February 28, 2019). Noble gas. Encyclopædia Britannica. Recovered from: britannica.com



Yet No Comments