
Gases properties, behavior, shape, examples

The gases They are all those substances or compounds whose states of aggregation are weak and dispersed, at the same time that they depend enormously on the pressure and temperature conditions that govern them. They are perhaps the second most abundant form of matter in the entire Universe after plasma..
On Earth, gases make up the layers of the atmosphere, from the exosphere to the troposphere and the air we breathe. Although a gas is invisible when it is diffused through large spaces, such as the sky, it is detected by the movement of clouds, the turns of the blades of a mill, or by the vapors exhaled from our mouths in cold climates..

Likewise, going to the negative environmental aspects, it is observed in the black smoke from the exhaust pipes of the vehicles, in the smoke columns of the towers located in the factories, or in the smoke raised when a forest burns.
You are also facing gaseous phenomena when you see vapors that come out of the sewers, in the wisps of swamps and cemeteries, in the bubbling inside the fish tanks, in the helium balloons that are released into the sky, in the oxygen released by plants as a result of their photosynthesis, and even in belching and flatulence.
Wherever the gases are observed means that there was a chemical reaction, unless they are fixed or assimilated directly from the air, the main source of gases (superficially) on the planet. As temperatures rise, all substances (chemical elements) will transform into gases, including metals such as iron, gold, and silver..
Regardless of the chemical nature of gases, they all share in common the great distance that separates their particles (atoms, molecules, ions, etc.), which move chaotically and arbitrarily through a given volume or space..
Article index
- 1 Properties of gases
- 1.1 Physical
- 1.2 Electrical and thermal conductivity
- 1.3 Reactivity
- 2 Behavior of gases
- 3 Form of gases
- 4 Pressure of a gas
- 4.1 Units
- 5 Volume of a gas
- 6 Main gas laws
- 6.1 Boyle's law
- 6.2 Charles's Law
- 6.3 Gay-Lussac's Law
- 6.4 Avogadro's Law
- 7 Types of gases
- 7.1 Combustible gases
- 7.2 Industrial gases
- 7.3 Inert gases
- 8 Examples of gaseous elements and compounds
- 8.1 Gaseous compounds
- 9 References
Properties of gases
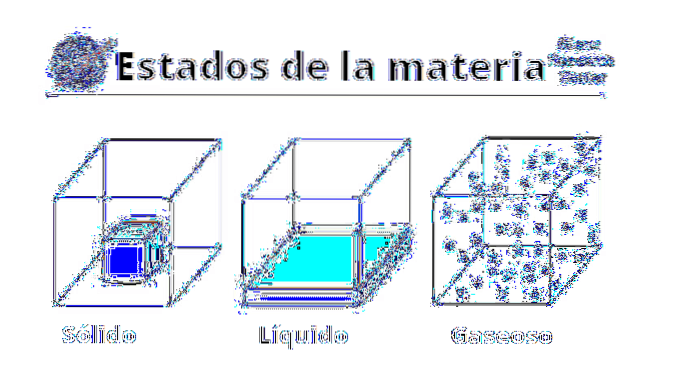
Physical
The physical properties of gases vary depending on what substance or compound it is. Gases are popularly associated with bad odors or putrefaction, either due to their sulfur content, or to the presence of volatile amines. Likewise, they are visualized with greenish, brown or yellowish colorations, which intimidate and give a bad omen.
However, most gases, or at least the most abundant ones, are actually colorless and odorless. Although they are elusive, they can be felt on the skin and resist movement, even creating viscous layers on the bodies that pass through them (as happens with airplanes)..
All gases can undergo pressure or temperature changes that end up turning them into their respective liquids; that is, they suffer condensation (if they are cooled) or liquefaction (if they are “pressed”).

On the other hand, gases are capable of dissolving in liquids and some porous solids (such as activated carbon). Bubbles are the result of accumulations of gases that have not yet dissolved in the medium and escape towards the surface of the liquid..
Electrical and thermal conductivity
Under normal conditions (without ionization of their particles), gases are poor conductors of heat and electricity. However, when they are stressed with many electrons, they allow current to pass through them, as seen in lightning during storms..
On the other hand, at low pressures and subjected to an electric field, some gases, especially the noble or perfect ones, light up and their lights are used for the design of night-time advertisements and posters (neon light), as well as in the famous electric discharge lamps in street lanterns.
Regarding thermal conductivity, many gases behave as thermal insulators, so their incorporation in the filling of fibers, fabrics, or glass panels, helps to prevent heat from passing through them and keeps the temperature constant..
However, there are gases that are good conductors of heat, and can cause worse burns than those caused by liquids or solids; for example, as happens with hot steam from baked cupcakes (or empanadas), or with jets of steam escaping from boilers.
Reactivity
Generally, reactions that involve gases, or where they occur, are classified as dangerous and cumbersome.
Their reactivity depends, again, on their chemical nature; However, when expanding and mobilizing with great ease, greater care and control must be exercised because they can trigger drastic increases in pressure that put the structure of the reactor at risk; not to mention how flammable or not blissful gases are.
Behavior of gases
Macroscopically one can get an idea of the behavior of gases by witnessing how smoke, rings, or the literary "tongues" of cigarettes evolve in the air. Likewise, when a smoke grenade explodes, it is interesting to detail the movement of these different colored clouds.
However, such observations are subject to the action of the air, and also to the fact that there are very fine solid particles suspended in the smoke. Therefore, these examples are not enough to reach a conclusion regarding the true behavior of a gas. Instead, experiments have been carried out and the kinetic theory of gases developed..
Molecularly and ideally, gaseous particles elastically collide with each other, having linear, rotational and vibrational displacements. They have an associated average energy, which allows them to travel freely through any space without almost interacting or colliding with another particle as the volume around them increases..
Its behavior would be a mixture of the erratic Brownian movement, and that of some collisions of some billiard balls that bounce incessantly between each other and the walls of the table; if there are no walls, they will diffuse towards infinity, unless they are held back by a force: gravity.
Form of gases
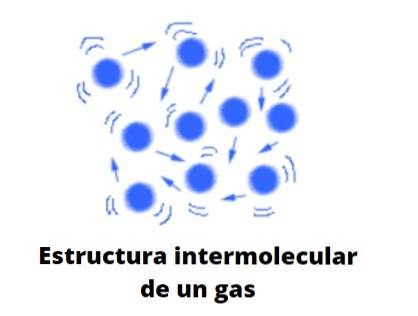
Gases, unlike liquids and solids, are not matter of the condensed type; that is, the aggregation or cohesion of its particles never manages to define a shape. They share with liquids the fact that they completely occupy the volume of the container that contains them; however, they lack surface and surface tension.
If the gas concentration is high, its “tongues” or the macroscopic forms already described can be seen with the naked eye. These, sooner or later, will end up vanishing by action of the wind or by the mere expansion of the gas. The gases therefore cover all the corners of the limited space originating highly homogeneous systems.
Now, the theory conveniently considers gases as spheres that hardly collide with themselves; but when they do, they bounce elastically.
These spheres are widely separated from each other, so the gases are practically "full" of vacuum; hence its versatility to pass through the slightest slit or fissure, and the ease of being able to significantly compress them.
That is why, no matter how closed a bakery installation is, if you walk next to it it is sure that you will enjoy the aroma of freshly baked breads..
Pressure of a gas
It could be believed that because the gas spheres or particles are so dispersed and separated, they are incapable of generating any pressure on the bodies or objects. However, the atmosphere proves that such a belief is false: it has mass, weight, and prevents liquids from evaporating or boiling out of nowhere. Boiling points are measured at atmospheric pressure.
Gas pressures become more quantifiable if manometers are available, or if they are enclosed in containers with non-deformable walls. Thus, the more gas particles there are inside the container, the greater the number of collisions between them and the walls of the container..
These particles, when they collide with the walls, press them, since they exert a force proportional to their kinetic energy on their surface. It is as if the ideal billiard balls were thrown at a wall; if there are many that impact them at high speed, it could even break.
Units
There are many units that accompany gas pressure measurements. Some of the best known are millimeters of mercury (mmHg), like torr. There are those of the international system of units (SI) that define the pascal (Pa) in terms of N / mtwo; and of him, the kilo (kPa), mega (MPa) and giga (GPa) pascal.
Volume of a gas
A gas occupies and expands throughout the volume of the container. The larger the container, the volume of the gas will be too; but both its pressure and density will decrease for the same amount of particles.
The gas itself, on the other hand, does have an associated volume that does not depend so much on its nature or molecular structure (ideally), but on the pressure and temperature conditions that govern it; this is, its molar volume.
In reality, the molar volume varies from one gas to another, although the variations are small if they are not heterogeneous and large molecules. For example, the molar volume of ammonia (NH3, 22,079 L / mol) at 0 ºC and 1 atm, differs from helium (He, 22,435 L / mol).
All gases have a molar volume that changes as a function of P and T, and no matter how large their particles are, the number of them is always the same. From here in fact he derived what is known by Avogadro's number (NTO).
Main gas laws
The behavior of gases has been studied for centuries through experiments, in-depth observations and interpretation of the results..
Such experiments made it possible to establish a series of laws that, put together in the same equation (that of ideal gases), help to predict the responses of a gas to different conditions of pressure and temperature. In this way, there is a relationship between its volume, temperature and pressure, as well as the number of its moles in a given system..
Among these laws are the following four: Boyle, Charles, Gay-Lussac and Avogadro's.
Boyle's Law
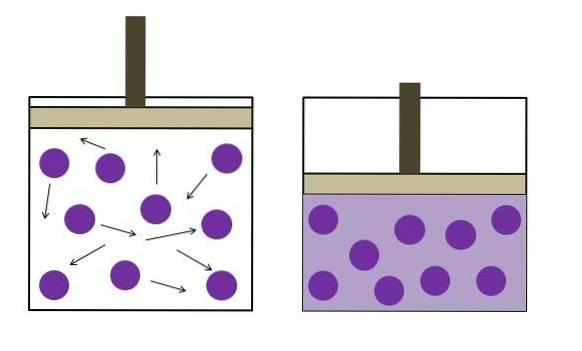
Boyle's law states that at constant temperature, the volume of an ideal gas is inversely proportional to its pressure; that is, the larger the container, the lower the pressure that its walls will experience due to the same amount of gas..
Charles Law

Charles's law states that at constant pressure, the volume of an ideal gas is directly proportional to its temperature. Balloons demonstrate Charles's law, because if they are heated they inflate a little more, while if they are submerged in liquid nitrogen, they deflate because the volume of gas inside them contracts..
Gay-Lussac Law
Gay-Lussac's law states that at constant volume, the pressure of an ideal gas is directly proportional to its temperature. In a well-closed cauldron, if a gas is heated progressively, each time the pressure inside it will be greater, because the walls of the cauldron do not deform or expand; that is, its volume does not change, it is constant.
Avogadro's Law
Finally, Avogadro's law states that the volume occupied by an ideal gas is directly proportional to the number of its particles. Thus, if we have one mole of particles (6.02 · 102. 3), we will then have the molar volume of the gas.
Types of gases
Combustible gases
They are those gases whose components work as fuels, because they are used for the production of thermal energy. Some of them are natural gas, liquefied petroleum gas and hydrogen.
Industrial gases
They are those manufactured gases, which are marketed to the public for different uses and applications, such as for the health, food, environmental protection, metallurgy, chemical industry, security sectors, among others. Some of these gases are oxygen, nitrogen, helium, chlorine, hydrogen, carbon monoxide, propane, methane, nitrous oxide, among others..
Inert gases
They are those gases that under specific temperature and pressure conditions, do not generate any chemical reaction or a very low one. They are neon, argon, helium, krypton, and xenon. They are used in chemical processes in which non-reactive elements are necessary.
Examples of gaseous elements and compounds
What are the gaseous elements of the periodic table under earth conditions?
We first have hydrogen (H), which forms H moleculestwo. Helium (He), the lightest noble gas, follows; and then nitrogen (N), oxygen (O) and fluorine (F). These last three also form diatomic molecules: Ntwo, ORtwo and Ftwo.
After fluorine comes neon (Ne), the noble gas that follows helium. Below fluorine we have chlorine (Cl), in the form of Cl moleculestwo.
Next we have the rest of the noble gases: argon (Ar), krypton (Kr), xenon (Xe), radon (Rn) and oganeson (Og).
Therefore, they are a total of twelve gaseous elements; eleven if we exclude the highly radioactive and unstable oganeson.
Gaseous compounds
In addition to gaseous elements, some common gaseous compounds will be listed:
-HtwoYes, hydrogen sulfide, responsible for the smell of rotten eggs
-NH3, ammonia, that pungent aroma found in used soaps
-COtwo, carbon dioxide, a greenhouse gas
-NOTtwo, nitrogen dioxide
-NO, nitrogen monoxide, a gas that was believed to be highly toxic but plays an important role in the circulatory system
-SW3, sulfur trioxide
-C4H10, butane
-HCl, hydrogen chloride
-OR3, ozone
-SF6, sulfur hexafluoride
References
- Whitten, Davis, Peck & Stanley. (2008). Chemistry. (8th ed.). CENGAGE Learning.
- The Properties of Gases. Recovered from: chemed.chem.purdue.edu
- Wikipedia. (2019). Gas. Recovered from: en.wikipedia.org
- Helmenstine, Anne Marie, Ph.D. (December 05, 2018). Gases - General Properties of Gases. Recovered from: thoughtco.com
- Harvard Men's Health Watch. (2019). The state of gas. Recovered from: health.harvard.edu
- Electronics Cooling Editors. (September 1, 1998). The thermal conductivity of gases. Recovered from: electronics-cooling.com


Yet No Comments