
Glutathione characteristics, structure, functions, biosynthesis
The glutathione (GSH) is a small tripeptide molecule (with only three amino acid residues) non-protein that participates in many biological phenomena such as enzymatic mechanics, macromolecule biosynthesis, intermediate metabolism, oxygen toxicity, intracellular transport, etc..
This small peptide, present in animals, plants and some bacteria, is considered a "buffer" oxido-reducing, since it is one of the main low molecular weight compounds that contains sulfur and lacks the toxicity associated with cysteine residues.
Some diseases in humans have been associated with the deficiency of specific enzymes of glutathione metabolism, and this is due to its multiple functions in maintaining body homeostasis.
Malnutrition, oxidative stress and other pathologies suffered by humans can be evidenced as a drastic decrease in glutathione, which is why it is sometimes a good indicator of the state of health of the body systems.
For plants, in the same way, glutathione is an essential factor for their growth and development, since it also fulfills functions in multiple biosynthetic pathways and is essential for cellular detoxification and internal homeostasis, where it acts as a powerful antioxidant..
Article index
- 1 Features
- 2 Structure
- 3 Functions
- 3.1 Coenzyme
- 3.2 Storage of cysteine
- 3.3 Protein folding
- 3.4 Function in erythrocytes
- 3.5 Xenobiotic metabolism
- 3.6 Oxidative state of cells
- 4 Biosynthesis
- 4.1 Reduced glutathione (GSH)
- 4.2 Oxidized glutathione (GSSG)
- 5 Benefits of its intake
- 5.1 Cancer
- 5.2 HIV
- 5.3 Muscle activity
- 5.4 Liver pathologies
- 5.5 Antioxidant
- 5.6 "Absorption"
- 6 Side effects
- 7 References
Characteristics
The first studies carried out in relation to the subcellular location of glutathione showed that it is present in the mitochondria. Later, it was also observed in the region corresponding to the nuclear matrix and in peroxisomes.
At present it is known that the compartment where its concentration is most abundant is in the cytosol, since it is actively produced there and is transported to other cellular compartments such as the mitochondria..
In mammalian cells, the concentration of glutathione is in the millimole range, while in blood plasma its reduced form (GSH) is found in micromolar concentrations..
This intracellular concentration closely resembles the concentration of glucose, potassium, and cholesterol, essential elements for cellular structure, function, and metabolism..
Some organisms have analogous or variant glutathione molecules. Protozoan parasites that affect mammals have a form known as "trypanothion" and in some bacteria this compound is replaced by other sulfurized molecules such as thiosulfate and glutamylcysteine..
Certain plant species have, in addition to glutathione, homologous molecules that have residues other than glycine at the C-terminal end (homoglutathione), and that are characterized by having functions similar to those of the tripeptide in question..
Despite the existence of other compounds similar to glutathione in different organisms, this is one of the "thiols" found in the highest concentration intracellularly..
The high ratio that normally exists between the reduced form (GSH) and the oxidized form (GSSG) of glutathione is another distinguishing feature of this molecule..
Structure
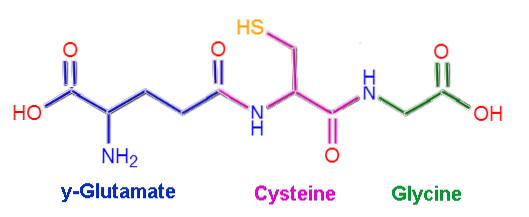
Glutathione or L-γ-glutamyl-L-cysteinyl-glycine, as its name implies, is composed of three amino acid residues: L-glutamate, L-cysteine and glycine. Cysteine and glycine residues are linked together through common peptide bonds, that is, between the α-carboxyl group of one amino acid and the α-amino group of the other..
However, the bond that occurs between glutamate and cysteine is not typical of proteins, since it occurs between the γ-carboxyl portion of the R group of glutamate and the α-amino group of cysteine. is called γ bond.
This small molecule has a molar mass of just over 300 g / mol and the presence of the γ bond appears to be crucial for the immunity of this peptide against the action of many aminopeptidase enzymes..
Features
As mentioned, glutathione is a protein that participates in numerous cellular processes in animals, plants, and certain prokaryotes. In this sense, its general participation in:
-The processes of protein synthesis and degradation
-The formation of DNA ribonucleotide precursors
-Regulation of the activity of some enzymes
-Protection of cells in the presence of reactive oxygen species (ROS) and other free radicals
-Signal transduction
-Genetic expression and in
-Apoptosis or programmed cell death
Coenzyme
It has also been determined that glutathione functions as a coenzyme in many enzymatic reactions, and that part of its importance is related to its ability to transport amino acids in the form of γ-glutamyl amino acids intracellularly..
The glutathione that can leave the cell (which it does in its reduced form) is capable of participating in oxidation-reduction reactions in the vicinity of the plasma membrane and the surrounding cell environment, which protects cells from damage against different classes of oxidizing agents.
Cysteine storage
This tripeptide also functions as a cysteine storage source and contributes to the maintenance of the reduced state of the sulfhydryl groups of the proteins inside the cell and the ferrous state of the heme group of the proteins that contain said cofactor.
Protein folding
When it participates in protein folding, it appears to have an important function as a reducing agent for disulfide bridges that have been formed inappropriately in protein structures, which is usually due to exposure to oxidizing agents such as oxygen, hydrogen peroxide, peroxynitrite, and some superoxides.
Erythrocyte function
In erythrocytes, the reduced glutathione (GSH) produced by the enzyme glutathione reductase, which uses the NADPH produced by the pentose phosphate pathway, contributes to the removal of hydrogen peroxide through the reaction catalyzed by another enzyme: glutathione peroxidase, which produces water and oxidized glutathione (GSSG).
The breakdown of hydrogen peroxide and, therefore, the prevention of its accumulation in the erythrocytes, prolongs the life time of these cells, since it avoids the oxidative damage that can occur in the cell membrane and that can end in hemolysis.
Xenobiotic metabolism
Glutathione is also an important player in the metabolism of xenobiotics, thanks to the action of glutathione S-transferase enzymes that generate glutathione conjugates that can then be metabolized intracellularly..
It is prudent to remember that the term "xenobiotic" is used to refer to drugs, environmental pollutants and chemical carcinogens to which an organism is exposed..
Oxidative state of cells
Since glutathione exists in two forms, one reduced and one oxidized, the relationship between the two molecules determines the redox state of cells. If the GSH / GSSG ratio is greater than 100, the cells are considered healthy, but if it is close to 1 or 10, it can be an indicator that the cells are in a state of oxidative stress.
Biosynthesis
The glutathione tripeptide is synthesized inside cells, both in plants and animals, by the action of two enzymes: (1) γ-glutamylcysteine synthetase and (2) glutathione synthetase (GSH synthetase), while its degradation or " decomposition ”depends on the action of the enzyme γ-glutamyl transpeptidase.
In plant organisms, each of the enzymes is encoded by a single gene and defects in any of the proteins or their encoding genes can cause lethality in embryos.
In humans, as in other mammals, the main site of glutathione synthesis and export is the liver, specifically in liver cells (hepatocytes) that surround the venous ducts that transport blood and other substances to and from the organ in question.
Synthesis de novo glutathione, its regeneration or recycling, require energy from ATP to occur.
Reduced glutathione (GSH)
Reduced glutathione is derived from the amino acids glycine, glutamate and cysteine, as already mentioned, and its synthesis begins with the activation (using ATP) of the γ-carboxyl group of glutamate (of the R group) to form an intermediate acyl phosphate, which is attacked by the α-amino group of cysteine.
This first two amino acid condensation reaction is catalyzed by γ-glutamylcysteine synthetase and is usually affected by the intracellular availability of the amino acids glutamate and cysteine..
The dipeptide thus formed is subsequently condensed with a glycine molecule thanks to the action of GSH synthetase. During this reaction, an activation with ATP of the α-carboxyl group of cysteine also occurs to form an acyl phosphate and thus favor the reaction with the glycine residue..
Oxidized glutathione (GSSG)
When reduced glutathione participates in oxidation-reduction reactions, the oxidized form is actually made up of two glutathione molecules linked together through disulfide bridges; it is for this reason that the oxidized form is abbreviated with the acronym "GSSG".
The formation of the oxidized species of glutathione depends on an enzyme known as glutathione peroxidase or GSH peroxidase, which is a peroxidase that contains a selenocysteine (a cysteine residue that instead of having a sulfur atom has a selenium atom) in place. active.
The interconversion between the oxidized and reduced forms occurs thanks to the participation of a GSSG reductase or glutathione reductase, which uses NAPDH to catalyze the reduction of GSSG in the presence of oxygen, with the concomitant formation of hydrogen peroxide.
Benefits of its intake
Glutathione can be administered orally, topically, intravenously, intranasally or nebulized, in order to increase its systemic concentration in patients suffering from oxidative stress, for example.
Cancer
Research on the oral administration of glutathione suggests that its intake may reduce the risks of oral cancer and that, when administered in conjunction with oxidative chemotherapeutics, it reduces the negative effects of the therapy in cancer patients..
HIV
Generally, patients infected with the acquired immunodeficiency virus (HIV) have intracellular glutathione deficiencies in both red blood cells and T cells and monocytes, which determines their correct functioning.
In a study by Morris et al., It was shown that the supply of glutathione to macrophages from HIV-positive patients considerably improved the function of these cells, especially against infections with opportunistic pathogens such as M. tuberculosis.
Muscle activity
Other studies have to do with the improvement of muscle contractile activity, antioxidative defense and oxidative damage caused in response to ischemia / reperfusion injuries after oral administration of GSH during physical resistance training.
Liver pathologies
It has been considered, in turn, that its ingestion or intravenous administration has functions in the prevention of the progress of some types of cancer and in the reduction of cellular damage that occurs as a result of certain liver diseases.
Antioxidant
Although not all the studies reported have been carried out in human patients, but are usually tests in animal models (generally murine), the results obtained in some clinical trials confirm the effectiveness of exogenous glutathione as an antioxidant..
For this reason, it is used for the treatment of cataracts and glaucoma, as an "anti-aging" product, for the treatment of hepatitis, numerous heart diseases, memory loss and to strengthen the immune system, and for the Clearance after heavy metal and drug poisoning.
"Absorption"
Exogenously administered glutathione cannot enter cells unless it is hydrolyzed to its constituent amino acids. Therefore, the direct effect of the administration (oral or intravenous) of this compound is the increase of the intracellular concentration of GSH thanks to the contribution of the amino acids necessary for its synthesis, which can be effectively transported to the cytosol.
Side effects
Although glutathione intake is considered to be "safe" or harmless, not enough studies have been done on its side effects..
However, from the few studies reported it is known that it can have negative effects that result from the interaction with other drugs and that can be harmful to health in various physiological contexts.
If taken long-term, it seems that they act in excessively lowering zinc levels and, in addition, if inhaled, it can produce severe asthma attacks in asthmatic patients..
References
- Allen, J., & Bradley, R. (2011). Effects of Oral Glutathione Supplementation on Systemic Oxidative Stress Biomarkers in Human Volunteers. The Journal of Alternative and Complementary Medicine, 17(9), 827-833.
- Conklin, K. A. (2009). Dietary Antioxidants During Cancer Chemotherapy: Impact on Chemotherapeutic Effectiveness and Development of Side Effects. Nutrition and Cancer, 37(1), 1-18.
- Meister, A. (1988). Glutathione Metabolism and Its Selective Modification. The Journal of Biological Chemistry, 263(33), 17205-17208.
- Meister, A., & Anderson, M. E. (1983). Glutathione. Ann. Rev Biochem., 52, 711-760.
- Morris, D., Guerra, C., Khurasany, M., Guilford, F., & Saviola, B. (2013). Glutathione Supplementation Improves Macrophage Functions in HIV. Journal of Interferon & Cytokine Research, eleven.
- Murray, R., Bender, D., Botham, K., Kennelly, P., Rodwell, V., & Weil, P. (2009). Harper's Illustrated Biochemistry (28th ed.). McGraw-Hill Medical.
- Nelson, D. L., & Cox, M. M. (2009). Lehninger Principles of Biochemistry. Omega editions (5th ed.). https://doi.org/10.1007/s13398-014-0173-7.2
- Noctor, G., Mhamdi, A., Chaouch, S., Han, Y. I., Neukermans, J., Marquez-garcia, B.,… Foyer, C. H. (2012). Glutathione in plants: an integrated overview. Plant, Cell & Environment, 35, 454-484.
- Pizzorno, J. (2014). Glutathione! Investigative Medicine, 13(1), 8-12.
- Qanungo, S., Starke, D. W., Pai, H. V, Mieyal, J. J., & Nieminen, A. (2007). Glutathione Supplementation Potentiates Hypoxic Apoptosis by S-Glutathionylation of p65-NFkB. The Journal of Biological Chemistry, 282(25), 18427-18436.
- Ramires, P. R., & Ji, L. L. (2001). Glutathione supplementation and training increases myocardial resistance to ischemia-reperfusion in vivo. Ann. J. Physiol. Heart Circ. Physiol., 281, 679-688.
- Sies, H. (2000). Glutathione and its Role in Cellular Functions. Free Radical Biology & Medicine R, 27(99), 916-921.
- Wu, G., Fang, Y., Yang, S., Lupton, J. R., & Turner, N. D. (2004). Glutathione Metabolism and Its Implications for Health. American Society for Nutritional Sciences, 489-492.



Yet No Comments