
Guanine characteristics, structure, formation and functions
The guanine It is a nitrogenous base that serves for the biosynthesis of guanylate 5'-monophosphate and deoxyguanylate 5'-monophosphate. Both substances are part, respectively, of RNA and DNA, which store the genetic information of cells.
Ribonucleic acid (RNA) and deoxyribonucleic acid (DNA) are made up of nucleotides, which are made up of a nitrogenous base attached to a sugar and a phosphate group.
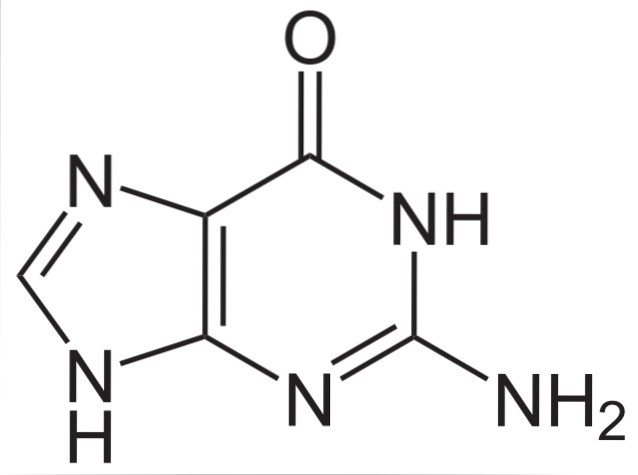
Guanine, in addition to being part of nucleic acids, in its forms of nucleosides monophosphate, diphosphate and triphosphates (GMP, GDP and GTP) participates in processes such as energy metabolism, the translation of the intracellular signal, the physiology of photoreceptors and vesicle fusion.
Article index
- 1 Chemical structure
- 2 Features
- 3 Biosynthesis
- 4 Function
- 5 Related diseases
- 5.1 Lesch-Nyhan syndrome
- 5.2 Increased PRPP synthase activity
- 5.3 von Gierke syndrome
- 6 References
Chemical structure
The chemical structure of guanine (2-amino-6-hydroxypurine) is a heterocyclic purine ring, consisting of a system of two linked rings: one ring is pyrimidine and the other ring is imidazole..
The heterocyclic ring of guanine is flat and with some conjugated double bonds. Additionally, it has two tautomeric forms, the keto and enol forms, between the C-1 and N-6 groups..
Characteristics
The characteristics of guanine are as follows:
- Guanine is an apolar substance. It is insoluble in water, but it is soluble in concentrated solutions of strong acids or bases.
- It can be isolated as a white solid, with empirical formula C5H5N5O, and molecular weight 151.3 g / mol.
- DNA's property of absorbing light at 260 nm is due, in part, to the chemical structure of guanine.
- In DNA, guanine forms three hydrogen bonds. The C-6 carbonyl group is a hydrogen bond acceptor, the N-1 group and the C-2 amino group are hydrogen bond donors.
For this reason, it takes more energy to break a bond between a guanine and a cytosine, than that of an adenine with a thymine, since the latter pair is linked by only two hydrogen bonds..
- In the cell, it is always found as part of nucleic acids or as GMP, GDP and GTP, never in its free form.
Biosynthesis
The guanine molecule, like other purines, is synthesized de novo from 5-phosphoribosyl-1-pyrophosphate (PRPP), through reactions catalyzed by enzymes.
The first step consists of the addition of an amino group, from glutamine, to PRPP and 5-phosphoribosylamine (PRA) is formed.
Subsequently, in an ordered sequence, the addition of glycine, aspartate, glutamine, formate, and carbon dioxide to the PRA occurs. In this way, an intermediate metabolite called inosine 5'-monophosphate (IMP) is formed..
During this process, free energy is used from the hydrolysis of ATP (adenosine 5'-triphosphate), which produces ADP (adenosine 5'-diphosphate) and Pi (inorganic phosphate)..
An oxidation of IMP is dependent on NAD+ (nicotinamide adenine dinucleotide), produces xanthine 5'-monophosphate (XMP). Subsequent addition of an amino group to XMP produces the guanylate molecule.
The regulation of guanylate biosynthesis takes place at the beginning, when PRA is formed, and at the end, when IMP oxidation occurs. Regulation occurs by negative feedback: a GMP nucleotide inhibits enzymes in both stages.
During the metabolic degradation of nucleotides, nitrogenous bases are recycled. GMP is formed by the enzyme hypoxanthine-guanine phosphoribosyltransferase, transferring a phosribosyl group from PRPP to guanine.
Function
Because guanine is not found in its free form, its functions are associated with GMP, GDP and GTP. Some of them are mentioned below:
- Guanosine 5'-triphosphate (GTP) acts as a reservoir of free energy. The gamma phosphate group of GTP can be transferred to adenosine 5'-triphosphate (ADP), to form ATP. This reaction is reversible, and is catalyzed by nucleoside diphosphate kinase..
- GMP is the most stable form of the nucleotide that contains guanine. Through hydrolysis, GMP forms cyclic GMP (cGMP), which is a second messenger during intracellular signaling, in the translation pathways. For example, in the photoreceptor and chemoreceptor cells of smell.
- CGMP participates in the relaxation of smooth muscle blood vessels during nitric oxide biosynthesis in endothelial cells.
- GTP gamma phosphate hydrolysis serves as a free energy source for protein biosynthesis in ribosomes.
- Hellicase enzymes require free energy from GTP hydrolysis to separate the DNA double helix, during DNA replication and transcription.
- In hippocampal neurons, the action of voltage-gated sodium channels is regulated by the hydrolysis of GTP to GDP.
Related diseases
High levels of uric acid in blood and urine have been associated with three different metabolic defects, which we will see below..
Lesch-Nyhan syndrome
It is characterized by a deficiency of HPRT (hypoxanthine-guanine phosphoribosyl transferase), an enzyme important for the recycling of hypoxanthine and guanine. In this case, PRPP levels increase and IMP and GMP, two important regulators of the initial stage of purine synthesis, are not formed. All this favors biosynthesis de novo of purines.
Increased PRPP synthase activity
This produces an increase in PRPP levels. This metabolite acts as an activator of glutamine PRPP-amidotransferase, which is responsible for the synthesis of 5-phosphoribosylamine, increasing biosynthesis de novo purines.
Von Gierke syndrome
It is a disease related to the storage of type I glycogen. Patients with this syndrome have a defective glucose 6-phosphatase. This produces an increase in glucose 6-phosphate levels, which serves for the synthesis of ribose 5-phosphate, via pentose phosphate..
Ribose 5-phosphate is the starting metabolite for PRPP biosynthesis. Similar to the two previous cases, this leads to an increase in biosynthesis de novo of purines.
Increased uric acid in the blood and urine causes symptoms that are commonly known as gout. In the case of Lesch Nyhan syndrome, patients completely lack the activity of the HPRP enzyme, leading to the manifestation of other symptoms including paralysis and mental retardation..
The HPRP gene is located on the X chromosome. Therefore, mutations in this gene affect males. There is no treatment to treat neurological problems. Symptoms associated with increased uric acid are treated with allopurinol.
References
- Dawson, R. et al. 1986. Data for Biochemical Research. Clarendon Press, Oxford.
- Horton, R; Moran, L; Scrimgeour, G; Perry, M. And Rawn, D. 2008. Principles of Biochemistry. 4th Edition. Pearson Education.
- Mathews, Van Holde, Ahern. 2001. Biochemistry. 3rd Edition.
- Murray, R; Granner, D; Mayes, P. And Rodwell, V. 2003. Harper's Illustrated Biochemistry. 26th Edition. McGraw-Hill Companies.
- Nelson, DL and Cox, M. 1994. Lehninger. Principles of Biochemistry. 4th Edition. Ed omega.
- Sigma-Aldrich. 2019. Guanine Chemical Sheet. Word Wide Web address: sigmaaldrich.com.



Yet No Comments