
Isopropyl characteristics, structure and examples
The isopropyl or isopropyl is an alkyl group or substituent, one of the most common and simple found in organic chemistry. It is one of the structural isomers of propyl, CH3CHtwoCHtwo-, group derived from propane, CH3CHtwoCH3. Its skeleton could be written as (CH3)twoCH-, showing that it also has three carbons.
Isopropyl when found in large molecules is simplified with the symbol iPr; although in small compounds it is not necessary and it is very easy to identify it. From it, a variety of organic compounds can be obtained that, although simple in appearance, are versatile for different applications; among them, isopropyl alcohol, (CH3)twoCHOH.

Shown above is the carbon skeleton of the isopropyl group. Note that in the center we have the “secondary” carbon, which is linked to two methyl groups, CH3, to a hydrogen and to an unknown segment represented by sinuosities; These can be a heteroatom, functional group, aliphatic chain (R), aromatic ring (Ar), etc..
If observed, the isopropyl group resembles a Y, a characteristic that is essential to recognize it at first glance when considering any molecular structure. The smaller the isopropyl is compared to the molecule of which it is a part, it is said that it acts simply as a substituent, which occurs in many compounds.
Article index
- 1 Features
- 2 Structure
- 3 Examples of compounds with isopropyl
- 3.1 Isopropyl alcohol
- 3.2 Isopropyl halides
- 3.3 Isopropylamine
- 3.4 Lorcainide
- 3.5 Others
- 4 References
Characteristics
Involving a little more the chemical nature of the isopropyl group, we can add that in addition to being alkyl (and aliphatic), that is, composed only of carbon and hydrogen atoms, it is nonpolar. This is because all its bonds, C-H and C-C, are of low polarity, so there are no regions rich or poor in electrons (dipoles).
The apolarity of isopropyl does not mean that it is electron poor; on the contrary, as a substituent it gives them to the more electronegative atoms. That is why wherever we see the Y in a molecule, we will know that it has given electron density to its surroundings; although the end result is to be an area lacking a dipole.
Isopropyl is also said to be aliphatic because it lacks aromaticity, found in rings with conjugated double bonds (benzene type).
Regarding its name, the origin of the iso- prefix will be explained in the following section. However, its IUPAC name comes to be 1-methylethyl, since in the second carbon of the ethyl chain, -CHtwoCH3, we substitute a H for a methyl group, -CH (CH3) CH3, which can also be written as -CH (CH3)two.
Structure
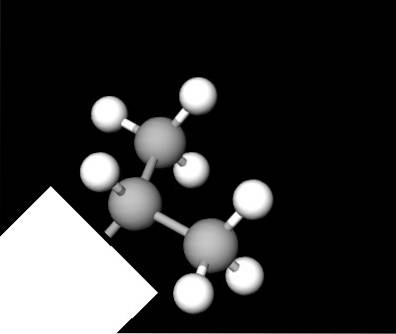
Above is the structure of the isopropyl group represented by a spheres and bars model. In it we again appreciate the Y; but it is not flat as one might think at first. The white box acts like the sinuosities of the first image, hiding the other segment of the molecule regardless of the compound..
All carbon atoms have chemical sp hybridization3, so it is not possible that they reside in the same plane. The C-C bonds have an approximate angle of 109'5º, which deforms the ends of the Y a little below or above its hypothetical plane..
This view of its structure is very useful because it makes it even easier to recognize when looking at other molecular models..
If a mirror plane were drawn in the middle of the CH carbon, it would be seen that the methyl groups are "reflected" on both sides of the mirror. Therefore, this plane divides the group -CH (CH3)two in two identical halves; a fact that would not happen with propyl, as it is straight chain. From there comes the prefix iso-, from 'equal', used for this alkyl substituent.
Examples of compounds with isopropyl
Isopropyl alcohol
Perhaps isopropyl alcohol is the most notable isopropyl derivative compound, since it is also the simplest secondary alcohol, widely marketed in plastic bottles within pharmacies as an antiseptic solution. Its structure is:
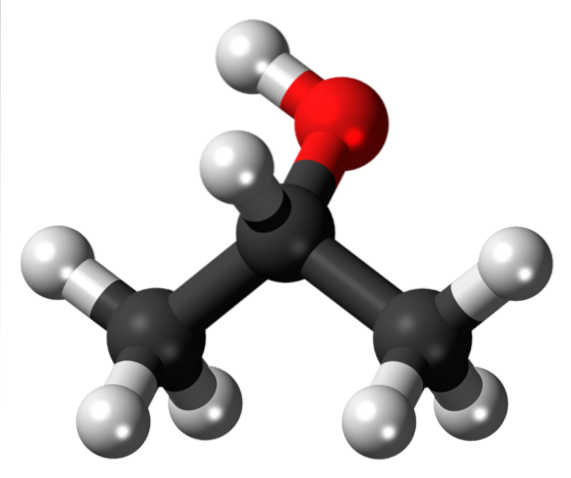
Note that in the center, under the OH group (red) we find the Y again, giving the false appearance of being flat. This alcohol is obtained simply by changing the sinuosities or the white box already shown by the OH group. The same applies for other groups or heteroatoms.
Isopropyl Halides
Suppose now it is not the OH but a halogen atom X (F, Cl, Br and I). In that case, we obtain the isopropyl halides, XCH (CH3)two. These organic compounds are characterized by being highly volatile liquids, possibly used as isopropyl sources in alkylation reactions (their addition to other molecules)..
Among these halides therefore we have:
-Isopropyl fluoride, FCH (CH3)two
-Isopropyl chloride, ClCH (CH3)two
-Isopropyl bromide, BrCH (CH3)two
-Isopropyl iodide, ICH (CH3)two
Its structures are identical to that of isopropyl alcohol, only changing the size of the atoms attached to the central or secondary carbon..
Isopropylamine
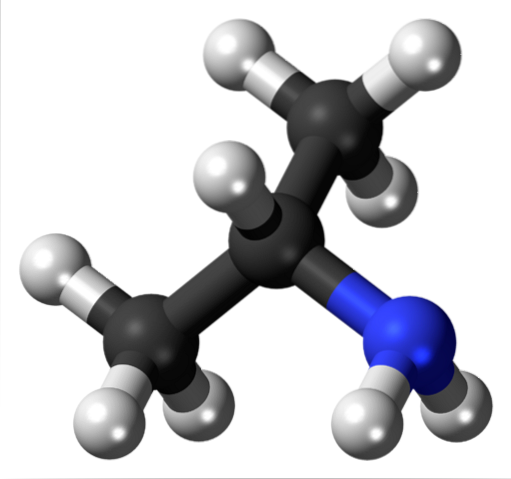
Now it is not about the OH or the halogens, but about the amino functional group, NHtwo (top image). As with isopropyl alcohol, isopropylamine is a secondary amine and a starting point for the synthesis of various insecticides. It is not marketed in the same way, since it is stinky and flammable, therefore being a dangerous compound.
Lorcainida

We left isopropyl derivatives behind and began to see it as a simple substituent. Lorcainide (above) is a controversial drug used to normalize heart rhythms. If the molecule is observed, it will be easy in a few seconds to see the Y to its left, which represents isopropyl.
In some formulas, the symbol iPr is used instead of Y; but it is usually applied for even more voluminous and complicated molecules.
Others
Finally, other examples with their respective formulas or structural models will be cited to serve as an exercise to locate isopropyl:

The Y of isopropyl is now located on the right side according to this representation of mazapertine, an antipsychotic drug.
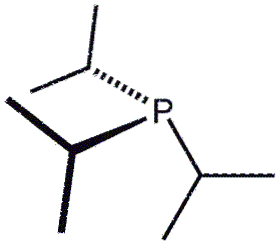
This time we have three isopropyl groups, P (CH (CH3)two)3, represented by three Ys. The formula could also be written as PiPr3 o P (iPr)3.
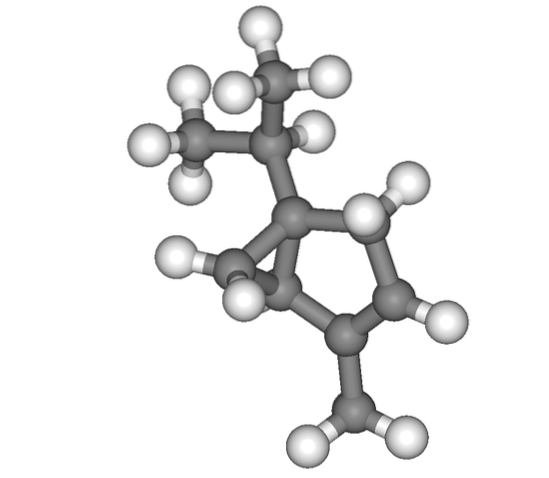
And finally we have the monoterpene thuyen, whose structure at first glance can cause confusion as to where the isopropyl is found. But after looking at it calmly, you will notice that this is at the top.
References
- Morrison, R. T. and Boyd, R, N. (1987). Organic chemistry. 5th Edition. Editorial Addison-Wesley Interamericana.
- Carey F. (2008). Organic chemistry. (Sixth edition). Mc Graw Hill.
- Graham Solomons T.W., Craig B. Fryhle. (2011). Organic chemistry. Amines. (10th edition.). Wiley plus.
- Steven A. Hardinger. (2017). Illustrated Glossary of Organic Chemistry: Isopropyl. Recovered from: chem.ucla.edu
- Elsevier B.V. (2019). Isopropyl Group. ScienceDirect. Recovered from: sciencedirect.com
- Wikipedia. (2019). Category: Isopropyl compounds. Recovered from: en.wikipedia.org


Yet No Comments