
Nucleoside characteristics, structure and applications
The nucleosides They are a large group of biological molecules made up of a nitrogenous base and a five-carbon sugar, covalently linked. In terms of structures they are very diverse.
They are the precursors for the synthesis of nucleic acids (DNA and RNA), a fundamental event for the control of the metabolism and growth of all living beings. They also participate in various biological processes, modulating some activities of the nervous, muscular and cardiovascular systems, among others..
Today, modified nucleosides are used as antiviral and anticancer therapy thanks to their property of blocking DNA replication.
It is important not to confuse the term nucleoside with nucleotide. Although both elements are structurally similar in that they are made up of the monomers of nucleic acids, the nucleotides have one or more additional phosphate groups. That is, a nucleotide is a nucleoside with a phosphate group.
Article index
- 1 Features
- 2 Structure
- 2.1 Nitrogen base
- 2.2 Pentose
- 2.3 Link
- 2.4 Modified nucleosides
- 3 Classification and nomenclature
- 4 Biological functions
- 4.1 Structural blocks
- 4.2 Energy storage
- 4.3 Local hormones
- 5 Nucleosides in the diet
- 6 Medical applications: anticancer and antiviral
- 7 References
Characteristics
Nucleosides are molecules made up of the building blocks of nucleic acids. They are of low molecular weight, being in a range between 227.22 to 383.31 g / mol.
Thanks to the nitrogenous base, these structures react as bases with pKa values between 3.3 and 9.8.
Structure
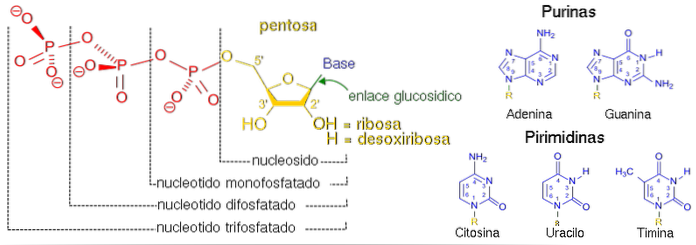
The nucleoside structure comprises a nitrogenous base linked by a covalent bond to a five-carbon sugar. We will explore these components thoroughly below..
Nitrogen base
The first component - the nitrogenous base, also called nucleobase - is a flat molecule of aromatic character that contains nitrogen in its structure, and can be a purine or a pyrimidine.
The former are made up of two fused rings: one of six atoms and the other of five. Pyrimidines are smaller and made up of a single ring.
Pentose
The second structural component is a pentose, which can be a ribose or a deoxyribose. Ribose is a "normal" sugar where each carbon atom is bonded with an oxygen atom. In the case of deoxyribose, the sugar is modified, since it lacks an oxygen atom at carbon 2 '.
Link
In all nucleosides (and also in nucleotides) that we find naturally, the bond between both molecules is of the β-N-glycosidic type, and it is resistant to alkaline cleavage..
The 1 'carbon of the sugar is attached to nitrogen 1 of pyrimidine and nitrogen 9 of purine. As we can see, these are the same components that we find in the monomers that make up nucleic acids: nucleotides.
Modified nucleosides
So far, we have described the general structure of nucleosides. However, there are some with certain chemical modifications, the most common being the union of a methyl group with the nitrogenous base. Methylations can also occur in the carbohydrate portion.
Other less frequent modifications include isomerization, for example from uridine to pseudouridine; loss of hydrogens; acetylation; formylation; and hydroxylation.
Classification and nomenclature
Depending on the structural components of the nucleoside, a classification into ribonucleosides and deoxynucleosides has been established. In the first category we find the nucleosides whose purine or pyrimidine is linked to a ribose. In addition, the nitrogenous bases that form them are adenine, guanine, cytosine and uracil.
In deoxynucleosides, the nitrogenous base is anchored to deoxyribose. The bases that we find are the same as in ribonucleotides, with the exception that pyrimidine uracil is replaced by a thymine.
In this way, ribonucleosides are named depending on the nitrogenous base that the molecule contains, establishing the following nomenclature: adenosine, cytidine, uridine and guanosine. To identify a deoxynucleoside, the prefix deoxy- is added, namely: deoxyadenosine, deoxycytidine, deoxyuridine and deoxyguanosine.
As we mentioned earlier, the fundamental difference between a nucleotide and a nucleoside is that the former has a phosphate group attached to the 3 'carbon (3'-nucleotide) or to the 5' carbon (5'-nucleotide). Thus, in terms of nomenclature, we can find that a synonym of the first case is a nucleoside-5'-phosphate.
Biological functions
Structural blocks
Nucleoside triphosphate (that is, with three phosphates in their structure) are the raw material for the construction of nucleic acids: DNA and RNA.
Energy storage
Thanks to the high-energy bonds that hold the phosphate groups together, they are structures that easily store energy of adequate availability for the cell. The most famous example is ATP (adenosine triphosphate), better known as the "energy currency of the cell".
Local hormones
The nucleosides themselves (without phosphate groups in their structure) do not have significant biological activity. However, in mammals we find a striking exception: the adenosine molecule.
In these organisms, adenosine takes on the role of autocoid, which means that it functions as a local hormone and also as a neuromodulator..
The circulation of adenosine in the bloodstream modulates different functions such as vasodilation, heart rate, contractions in the smooth muscles, the release of neurotransmitters, the degradation of lipids, among others..
Adenosine is famous for its role in regulating sleep. When the concentration of this nucleoside increases, it causes fatigue and sleep. That is why the consumption of caffeine (a molecule similar to adenosine) keeps us awake, since it blocks the interactions of adenosine and its respective receptors in the brain..
Nucleosides in the diet
Nucleosides can be consumed in food, and they have been shown to modulate various physiological processes, benefiting certain aspects of the immune system, development and growth of the gastrointestinal tract, lipid metabolism, liver functions, among others..
They are abundant components in breast milk, tea, beer, meat and fish, among other foods.
Exogenous nucleoside (and nucleotide) supplementation is important in patients who lack the ability to synthesize these compounds de novo.
Regarding absorption, almost 90% of the nucleotides are absorbed in the form of nucleosides and phosphorylated again in the cells of the intestine.
Medical applications: anticancer and antiviral
Certain nucleoside or modified nucleotide analogs have shown anticancer and antiviral activity, allowing the treatment of conditions of significant medical importance such as HIV / AIDS, herpes virus, hepatitis B virus, and leukemia, among others..
These molecules are used for the treatment of these pathologies, since they have the ability to inhibit DNA synthesis. These are actively transported into the cell and, as they present chemical modifications, prevent future replication of the virus genome..
The analogs that are used as treatment are synthesized through different chemical reactions. Modifications can come in the ribose portion or in the nitrogenous base.
References
- Alberts, B., Bray, D., Hopkin, K., Johnson, A. D., Lewis, J., Raff, M.,… & Walter, P. (2013). Essential cell biology. Garland science.
- Borea, P. A., Gessi, S., Merighi, S., Vincenzi, F., & Varani, K. (2018). Pharmacology of adenosine receptors: the state of the art. Physiological reviews, 98(3), 1591-1625.
- Cooper, G. M., & Hausman, R. E. (2007). The cell: a molecular approach. Washington, DC, Sunderland, MA.
- Griffiths, A. J. (2002). Modern genetic analysis: integrating genes and genomes. Macmillan.
- Griffiths, A. J., Wessler, S. R., Lewontin, R. C., Gelbart, W. M., Suzuki, D. T., & Miller, J. H. (2005). An introduction to genetic analysis. Macmillan.
- Koolman, J., & Röhm, K. H. (2005). Biochemistry: text and atlas. Panamerican Medical Ed..
- Mikhailopulo, I. A., & Miroshnikov, A. I. (2010). New trends in nucleoside biotechnology. Act Naturae 2(5).
- Passarge, E. (2009). Genetics text and atlas. Panamerican Medical Ed..
- Siegel, G. J. (1999). Basic neurochemistry: molecular, cellular and medical aspects. Lippincott-Raven.
Yet No Comments