
What is the heat of reaction?

The heat of reaction or Enthalpy of Reaction (ΔH) is the change in enthalpy of a chemical reaction that occurs at constant pressure. It is a thermodynamic unit of measure useful to calculate the amount of energy per mole that is released or produced in a reaction.
Since enthalpy is derived from pressure, volume, and internal energy, all of which are state functions, enthalpy is also a state function..
ΔH, or the enthalpy change emerged as a unit of measurement intended to calculate the energy change of a system when it became too difficult to find the ΔU, or change in the internal energy of a system, simultaneously measuring the amount of heat and work interchanged.
Given a constant pressure, the enthalpy change equals heat and can be measured as ΔH = q.
The notation ΔHº or ΔHºr then arises to explain the precise temperature and pressure of the heat of reaction ΔH.
The standard enthalpy of reaction is symbolized by ΔHº or ΔHºrxn and can assume both positive and negative values. The units for ΔHº are kiloJoules per mole, or kj / mol.
Previous concept to understand the heat of reaction: differences between ΔH and ΔHºr.
Δ = represents the change in enthalpy (enthalpy of products minus enthalpy of reactants).
A positive value indicates that the products have higher enthalpy, or that it is an endothermic reaction (heat is required).
A negative value indicates that the reactants have higher enthalpy, or that it is an exothermic reaction (heat is produced).
º = means that the reaction is a standard enthalpy change, and occurs at a preset pressure / temperature.
r = denotes that this change is the enthalpy of the reaction.
The Standard State: the standard state of a solid or liquid is the pure substance at a pressure of 1 bar or what is the same 1 atmosphere (105 Pa) and a temperature of 25 ° C, or what is the same 298 K.
The ΔHºr is the standard heat of reaction or standard enthalpy of a reaction, and as ΔH it also measures the enthalpy of a reaction. However, ΔHºrxn takes place under “standard” conditions, which means that the reaction takes place at 25º C and 1 atm..
The benefit of a ΔH measurement under standard conditions lies in the ability to relate one ΔHº value to another, since they occur under the same conditions.
Heat of formation
The standard heat of formation, ΔHFº, of a chemical product is the amount of heat absorbed or released from the formation of 1 mole of that chemical at 25 degrees Celsius and 1 bar of its elements in their standard states.
An element is in its standard state if it is in its most stable form and its physical state (solid, liquid or gas) at 25 degrees Celsius and 1 bar.
For example, the standard heat of formation for carbon dioxide involves oxygen and carbon as reactants..
Oxygen is more stable as O gas moleculestwo, while carbon is more stable as solid graphite. (Graphite is more stable than diamond under standard conditions).
To express the definition another way, the standard heat of formation is a special type of standard heat of reaction..
The reaction is the formation of 1 mole of a chemical from its elements in their standard states under standard conditions.
The standard heat of formation is also called the standard enthalpy of formation (although it is actually a change in enthalpy).
By definition, the formation of an element of itself would not produce any change in enthalpy, so the standard heat of reaction for all elements is zero (Cai, 2014).
Calculation of reaction enthalpy
1- Experimental calculation
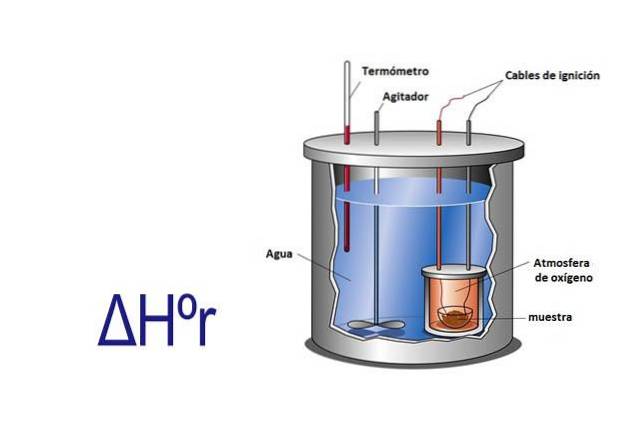
Enthalpy can be measured experimentally by using a calorimeter. A calorimeter is an instrument where a sample is reacted through electrical cables that provide the activation energy. The sample is in a container surrounded by water which is constantly stirred.
By measuring with a the change in temperature that occurs when the sample reacts, and knowing the specific heat of water and its mass, the heat released or absorbed by the reaction is calculated using the equation q = Cesp x m x ΔT.
In this equation q is heat, Cesp is the specific heat in this case of water which is equal to 1 calorie per gram, m is the mass of water and ΔT is the change in temperature.
The calorimeter is an isolated system that has a constant pressure, so ΔHr= q
2- Theoretical calculation
The enthalpy change does not depend on the particular pathway of a reaction, but only on the global energy level of the products and reactants. Enthalpy is a function of state, and as such, it is additive.
To calculate the standard enthalpy of a reaction, we can add the standard enthalpies of formation of the reactants and subtract it from the sum of the standard enthalpies of formation of the products (Boundless, S.F.). Stated mathematically, this gives us:
ΔHr° = Σ ΔHFº (products) - Σ ΔHFº (reactants).
Enthalpies of reactions are usually calculated from enthalpies of reactant formation under normal conditions (pressure of 1 bar and temperature 25 degrees Celsius).
To explain this principle of thermodynamics, we will calculate the enthalpy of the reaction for the combustion of methane (CH4) according to the formula:
CH4 (g) + 2Otwo (g) → COtwo (g) + 2HtwoO (g)
To calculate the standard enthalpy of the reaction, we need to find the standard enthalpies of formation for each of the reactants and products involved in the reaction..
These are usually found in an appendix or in various tables online. For this reaction, the data we need are:
HFº CH4 (g) = -75 kjoul / mol.
HFº Otwo (g) = 0 kjoul / mol.
HFº COtwo (g) = -394 kjoul / mol.
HFº HtwoO (g) = -284 kjoul / mol.
Note that because it is in its standard state, the standard enthalpy of formation for oxygen gas is 0 kJ / mol.
Here we summarize our standard enthalpies of formation. Note that because the units are in kJ / mol, we need to multiply by the stoichiometric coefficients in the balanced reaction equation (Leaf Group Ltd, S.F.).
Σ ΔHFº (products) = ΔHFº COtwo +2 ΔHFº HtwoOR
Σ ΔHFº (products) = -1 (394 kjoul / mol) -2 (284 kjoul / mol) = -962 kjoul / mol
Σ ΔHFº (reactants) = ΔHFº CH4 + ΔHFº Otwo
Σ ΔHFº (reactants) = -75 kjoul / mol + 2 (0 kjoul / mol) = -75 kjoul / mol
Now, we can find the standard enthalpy of the reaction:
ΔHr° = Σ ΔHFº (products) - Σ ΔHFº (reactants) = (- 962) - (- 75) =
ΔHr° = - 887kJ / mol.
References
- Anne Marie Helmenstine. (2014, June 11). Enthalpy of Reaction Definition. Recovered from thoughtco: thoughtco.com.
- (S.F.). Standard Enthalpy of Reaction. Recovered from boundless: boundless.com.
- Cai, E. (2014, March 11). standard heat of formation. Recovered from chemicalstatistician: chemicalstatistician.wordpress.com.
- Clark, J. (2013, May). Various enthalpy change definitions. Recovered from chemguide.co.uk: chemguide.co.uk.
- Jonathan Nguyen, G. L. (2017, February 9). Standard Enthalpy of Formation. Recovered from chem.libretexts.org: chem.libretexts.org.
- Leaf Group Ltd. (S.F.). How to Calculate Enthalpies of Reaction. Recovered from sciencing: sciencing.com.
- Rachel Martin, E. Y. (2014, May 7). Heat of Reaction. Recovered from chem.libretexts.org: chem.libretexts.org.



Yet No Comments