
Xanthoprotein reaction rationale, procedure, use

The xanthoprotein reaction is a chemical procedure used to determine the presence or absence of aromatic amino acids, such as tyrosine and tryptophan, which can be in free form or constituting soluble proteins, peptides or polypeptides.
It has also been used to detect toxic substances in the blood that contain a benzene group in their structure. This test is also known as the xanthoprotein Becher reaction..
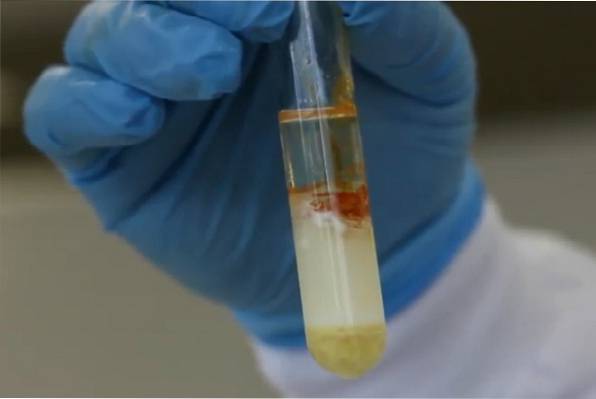
The xanthoprotein reaction uses concentrated nitric acid, heat, and a neutralizing alkali. If the solution turns from yellow to orange when the reaction is neutralized, the test is considered positive. The observed coloration is due to the formation of nitrogen compounds derived from the nitrification of benzene groups.
If the amount of total protein needs to be quantified, it is necessary to use other protein determination methods, such as Biuret..
The xanthoprotein reaction is used mainly when substances whose chemical composition is not known are being analyzed. This reaction is usually part of a set of tests that will help determine the chemical composition of a particular substance or extract. That is why it is widely used by researchers.
On the other hand, the Becher xanthoprotein reaction is used to detect phenolic and indole substances in the blood, making it possible to predict the appearance of nephropathy, before other later parameters are increased..
Article index
- 1 Rationale
- 2 Procedure
- 2.1 - Xanthoprotein reaction for the detection of amino acids with aromatic groups
- 2.2 - Xanthoprotein reaction to detect phenolic and indole substances in the blood
- 3 Use
- 4 Precautions
- 5 References
Basis
The reaction basically detects the presence of the benzene group, both in amino acids, as well as in proteins and peptides. Nitric acid acts on the benzene ring of amino acids that possess it, forming phenolic nitro compounds.
The reaction needs heat to be carried out, generally a precipitate forms that clouds the medium, turning it milky. The precipitate can be white or yellow. Finally, it requires an alkalization step that accentuates the color. For this, a base such as 40% sodium hydroxide or ammonia is used..
The reaction will be interpreted as positive if a strong yellow coloration is observed during the heating process or dark orange when the reaction alkalizes. The amino acids it detects are mainly tyrosine and tryptophan.
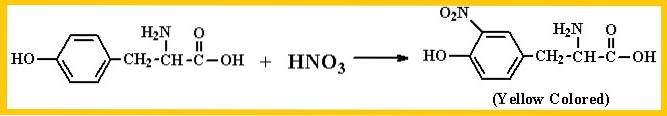
In the case of phenylalanine, although it is an aromatic amino acid, the benzene nucleus of this amino acid is difficult to be nitrated in a simple reaction, therefore, it will emit either a negative or weak positive reaction.
In general, the xanthoprotein test can be supplemented with the Hopkins-Cole test, which is used to specifically identify tryptophan. If the xanthoprotein test is positive and the Hopkins-Cole test is negative, the presence of tryptophan in that solution is ruled out, pointing to the presence mainly of tyrosine. Likewise, it can also be complemented with the Millon reaction for the detection of tyrosine, among other things..
This reaction can also be used on blood, plasma or desalbuminated serum samples for the diagnosis of uremia or renal failure. In this case, what is sought is the detection of toxic substances (phenolic and indole) produced from intestinal putrefaction..
These substances have the benzene group and therefore give a positive xanthoprotein reaction, although the reacting substance is neither an amino acid nor a protein..
Process
- Xanthoprotein reaction for the detection of amino acids with aromatic groups
Place 1 ml of the test sample in a clean, dry test tube.
-Add 0.5 ml of concentrated nitric acid.
-Incubate the mixture in a water bath at 70 ° C for 2 minutes. Previously prepare the water bath at the mentioned temperature.
-When removing the tube from the water bath, it is possible to observe that the solution has turned milky and has taken on a certain yellowish-white color..
-The solution is cooled by dropping cold water at the base of the tube..
-The preparation is made alkaline by slowly adding (drop by drop) a 40% sodium hydroxide solution until there is a color change.
-If the test is positive, a dark orange ring will form at the interface of the liquids..
-If the reaction is negative there will be no color formation.
- Xanthoprotein reaction to detect phenolic and indole substances in the blood
-Dispense 5 cc of 20% trichloroacetic acid on 5 cc of venous blood, plasma or serum, the latter being the most indicated. Mix well and filter.
-2 ml of the filtrate are taken and mixed with 0.5 ml of nitric acid. Then it is heated to boiling, for this the tube is placed directly over the lighter flame for 30 seconds. It is cooled with water and 1.5 of sodium chloride 33% is added..
It is interpreted as follows:
-Negative result: yellowish color.
-Positive result (+) lemon yellow.
-(++) golden yellow.
-(+++) dark yellow to brown
It can also be interpreted using a referential Autenrieth colorimetric scale, consisting of various concentrations of potassium dichromate. This is prepared in 10 in 10 dilutions from a 0.03874% stock solution. Each tube represents a percentage.
Interpretation scales
There are two scales of interpretation called ancient and modern. The normal values are:
Old table: between 75 to 85%
Modern table: between 15 to 25%
If the color of the test tube is in the middle of two standard tubes, an average is made between the two measurements.
The potassium dichromate standard solution is prepared as follows: 7.9 ml of 10 N potassium dichromate is measured and made up to 100 ml in a volumetric flask. For the dilutions, follow as shown in the Autenrieth colorimetric table..

Use
The use of the xanthoprotein reaction is part of a set of biochemical tests that help to know the chemical composition of some unknown substance. This test contributes to the chemical analysis the detection of proteins that contain benzene groups in their structure..
This reaction is widely used by researchers in various studies of substances of interest..
In this sense, Vásquez-Jorge and collaborators used this and other reactions to investigate the protein content of fluids extracted from the mangrove oyster. (Crassostrea rhizophorae).
Likewise, Rojas et al. (2009) used a series of tests, including the xanthoprotein reaction, to analyze glycomacropeptides in ricotta whey precipitates..
On the other hand, the Becher xanthoprotein reaction was modified by Volhard to be used for other purposes. In this case, for the detection of phenolic and indole substances that can rise much earlier (weeks or months) before the levels of urea and residual nitrogen in the blood increase..
These toxic wastes are formed from the intestinal putrefaction of food.
These substances accumulate in the blood when they are not eliminated correctly, therefore, a positive xanthoprotein test can predict the early onset of renal failure proper or a state of uremia..
Depending on the intensity of the color, you can determine how advanced it is. In this case the test is quantitative.
In healthy people this test gives a slight yellow color, corresponding to the normal levels of tryptophan and tyrosine that may be in the blood.
While, in kidney patients the xanthoprotein reaction is strongly positive due to the presence of the following substances: phenol, indole, paracresol, aromatic oxyacids, among others.
Precautions
This test must be performed by trained professionals. All biosafety regulations must be considered, since the reagents used are corrosive acids and bases..
If by accident a few drops of nitric acid fall on the skin or on the nails, they will turn yellow, since both structures contain proteins. In addition, on the skin and mucous membranes it causes severe burns.
If skin contact occurs, the protocol to follow should be to wash the area immediately with soap and water for approximately 20 minutes..
If it splashes into the eyes, it should be washed with plenty of fresh water, trying to keep the eyes open..
References
- "Xanthoprotein reaction" Wikipedia, The Free Encyclopedia. 2 Sep 2019, 17:49 UTC. 21 Oct 2019, 20:31 wikipedia.org/
- Vásquez-Jorge Y, Guerra-Molina L, Quintana-Tamayo J, Ramírez-Arzuaga J, Fernando-Ballesteros R, Vásquez-Jorge Y. Physicochemical characterization and protein content of fluid extracts of mangrove oyster (Crassostrea rhizophorae). Cuban Rev of Chemistry, 2014; 26 (1): 66-74. Available in Redalyc.
- Rojas E, Valbuena E, Torres G, García A, Piñero M, Galindo L. Isolation and performance of GMP by precipitation of whey with trichloroacetic acid. Cient. (Maracaibo) 2009; 19 (3): 295-302. Available in: ve.scielo.
- Vásquez A. Xanthoprotein reaction. Spanish clinical journal. 1949; 35 (5): 326-327. Available at: Elsevier
- Gálvez I, Gárate O. Becher and Andrewes reactions in urinary surgery. Urology Service of the Spanish Hospital. Argentine journal of urology 395-404


Yet No Comments