
Surfactants and biosurfactants what it is for, examples and uses
A surfactant is a chemical compound capable of reducing the surface tension of a liquid substance, acting at an interface or contact surface between two phases, for example water-air or water-oil.
The term surfactant comes from the English word surfactant, which in turn is derived from the acronym of the expression surface active agent, which means in Spanish agent with interfacial or surface activity.
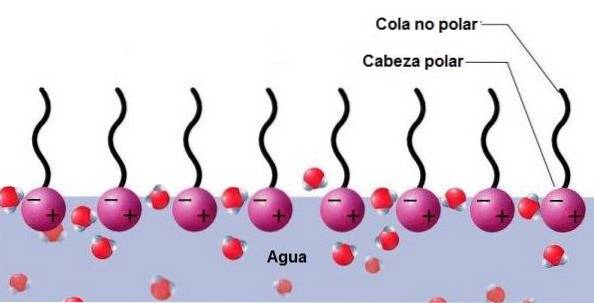
In Spanish, the word "surfactant" is used, referring to the ability of a chemical compound to act on surface or interfacial tension. Surface tension can be defined as a resistance that liquids have to increase their surface.
Water has a high surface tension because its molecules are very tightly bound and resist separating when pressure is exerted on their surface..
For example, some aquatic insects, such as "the cobbler" (Gerris lacustris), can move on the water without sinking, thanks to the surface tension of the water, which allows the formation of a film on its surface.

Also, a steel needle stays on the surface of the water and does not sink, due to the surface tension of the water..
Article index
- 1 Structure and function of surfactants
- 2 What are surfactants for?
- 3 Biosurfactants: surfactants of biological origin
- 3.1 Examples of biosurfactants
- 4 Classification of biosurfactants and examples
- 4.1 -Depending on the nature of the electrical charge in the polar part or head
- 4.2 -According to its chemical nature
- 4.3 -According to its molecular weight
- 5 Production of biosurfactants
- 6 Applications of biosurfactants
- 6.1 Oil industry
- 6.2 Environmental sanitation
- 6.3 In industrial processes
- 6.4 In the cosmetic and pharmaceutical industry
- 6.5 In the food industry
- 6.6 In agriculture
- 7 References
Structure and function of surfactants
All surfactant or surfactant chemical agents are of a nature amphiphilic, that is to say, they have a dual behavior, because they can dissolve polar and nonpolar compounds. Surfactants have two main parts in their structure:
- A hydrophilic polar head, akin to water and polar compounds.
- A lipophilic, hydrophobic nonpolar tail, akin to nonpolar compounds.
The polar head can be non-ionic or ionic. The surfactant tail, or apolar part, can be an alkyl or alkylbenzene carbon and hydrogen chain..
This very particular structure gives surfactant chemical compounds a dual, amphiphilic behavior: affinity for polar compounds or phases, soluble in water and also affinity for nonpolar compounds, insoluble in water..
In general, surfactant agents reduce the surface tension of water, which allows this liquid to expand and flow to a greater degree, wetting neighboring surfaces and phases..
What are surfactants for?
Surfactant chemicals exert their activity on surfaces or interfaces.
When dissolved in water, they migrate to the water-oil or water-air interfaces, for example, where they can function as:
- Dispersants and solubilizers of compounds insoluble or poorly soluble in water.
- Humectants, as they favor the passage of water to insoluble phases in this.
- Stabilizers for emulsions of compounds insoluble in water and water, such as oil and water from mayonnaise.
- Some surfactants promote and others prevent foaming.
Biosurfactants: surfactants of biological origin
When the surfactant comes from a living organism, it is called a biosurfactant..
In a stricter sense, biosurfactants are considered amphiphilic biological compounds (with dual chemical behavior, soluble in water and fat), produced by microorganisms such as yeast, bacteria and filamentous fungi..
Biosurfactants are excreted or retained as part of the microbial cell membrane.
Also some biosurfactants are produced by biotechnological processes, using enzymes that act on a biological chemical compound or natural product..
Examples of biosurfactants
Natural biosurfactants include saponins from plants such as cayenne flower (Hibiscus sp.), lecithin, mammalian bile juices, or human lung surfactant (with very important physiological functions).
Additionally, amino acids and their derivatives, betaines and phospholipids are biosurfactants, all of these natural products of biological origin..
Classification of biosurfactants and examples
-According to the nature of the electrical charge in the polar part or head
Biosurfactants can be grouped into the following categories, based on the electrical charge of their polar head:
Anionic biosurfactants
They have a negative charge at the polar end, frequently due to the presence of a sulphonate group -SO3-.
Cationic biosurfactants
They have a positive charge on the head, usually a quaternary ammonium group NR4+, where R represents a chain of carbon and hydrogen.
Amphoteric biosurfactants
They have both positive and negative charges on the same molecule.
Non-ionic biosurfactants
They do not have ions or electrical charges in their heads.
-According to its chemical nature
According to their chemical nature, biosurfactants are classified into the following types:
Glycolipid biosurfactants
Glycolipids are molecules that have in their chemical structure a part of lipid or fat and a part of sugar. Most of the known biosurfactants are glycolipids. The latter consist of sulfates of sugars such as glucose, galactose, mannose, rhamnose, and galactose..
Among the glycolipids, the best known are the rhamnolipids, bioemulsifiers that have been extensively studied, with high emulsifying activity and high affinity for hydrophobic organic molecules (which do not dissolve in water).
These are considered the most effective surfactants for the removal of hydrophobic compounds in contaminated soils..
Examples of rhamnolipids include surfactants produced by bacteria of the genus Pseudomonas.
There are other glycolipids, produced by Torulopsis sp., with biocidal activity and used in cosmetics, anti-dandruff products, bacteriostats and as body deodorants.
Lipoprotein and lipopeptide biosurfactants
Lipoproteins are chemical compounds that have in their structure a part of lipid or fat and another part of protein.
For example, Bacillus subtilis is a bacterium that produces lipopeptides called surfactins. These are one of the most powerful surface tension reducing biosurfactants..
Surfactins have the ability to produce erythrocyte lysis (breakdown of red blood cells) in mammals. In addition, they can be used as biocides for pests such as small rodents..
Fatty acid biosurfactants
Some microorganisms can oxidize alkanes (carbon and hydrogen chains) to fatty acids that have surfactant properties..
Phospholipid biosurfactants
Phospholipids are chemical compounds that have phosphate groups (PO43-), attached to a part with a lipid structure. They are part of the membranes of microorganisms.
Certain bacteria and yeasts that feed on hydrocarbons, when growing on alkane substrates, increase the amount of phospholipids in their membrane. For example, Acinetobacter sp., Thiobacillus thioxidans and Rhodococcus erythropolis.
Polymeric biosurfactants
Polymeric biosurfactants are high molecular weight macromolecules. The most studied biosurfactants in this group are: emulsify, liposan, mannoprotein and polysaccharide-protein complexes.
For example, the bacteria Acinetobacter calcoaceticus produces polyanionic emulsifier (with various negative charges), a very effective bioemulsifier for hydrocarbons in water. It is also one of the most powerful emulsion stabilizers known..
Liposan is an extracellular emulsifier, soluble in water, formed by polysaccharides and protein of Candida lipolytica.
Saccharomyces cereviseae produces large amounts of mannoproteins with excellent emulsifying activity for oils, alkanes and organic solvents.
-According to its molecular weight
Biosurfactants are classified into two categories:
Low molecular weight biosurfactants
With lower surface and interfacial tensions. For example, rhamnolipids.
High molecular weight polymeric biosurfactants
That bind strongly to surfaces, such as food bioemulsifiers.
Production of biosurfactants
Micro-organism cultures in bioreactors are used for the production of biosurfactants. Most of these microorganisms are isolated from contaminated environments, such as industrial waste sites or hydrocarbon pits disposed of by the oil industry..
The efficient production of biosurfactants depends on several factors, such as the nature of the substrate or carbon source used as the culture medium and its degree of salinity. In addition, it depends on factors such as temperature, pH and oxygen availability..
Applications of biosurfactants
Currently there is a huge commercial demand for biosurfactants, because the surfactants obtained by chemical synthesis (from petroleum derivatives) are toxic, non-biodegradable and therefore have environmental regulations for their use..
These issues have generated considerable interest in biosurfactants as non-toxic, biodegradable alternatives..
Biosurfactants have applications in many fields, such as:
Oil industry
Biosurfactants are used in oil extraction and bioremediation (decontamination with living organisms) of hydrocarbons; example: the biosurfactant of Arthrobacter sp.
They are also applied in biodesulfurization processes (removal of sulfur using microorganisms) from petroleum. Species of the genus have been used Rhodococcus.
Environmental sanitation
Biosurfactants are used in the bioremediation of soils contaminated by toxic metals, such as uranium, cadmium and lead (biosurfactants of Pseudomonas spp. Y Rhodococcus spp.).
They are also used in bioremediation processes of soils and water contaminated by spills of gasoline or oil..

For example, Aeromonas sp. produces biosurfactants that allow oil degradation or reduction of large molecules to smaller ones, which serve as nutrients for microorganisms, bacteria and fungi.
In industrial processes
Biosurfactants are used in the detergents and cleaners industry, since they enhance the cleaning action by dissolving the fats that dirty clothes or surfaces in the wash water..
They are also used as auxiliary chemical compounds in the textile, paper and tannery industries..
In the cosmetic and pharmaceutical industry
In the cosmetics industry, Bacillus licheniformis produces biosurfactants that are used as anti-dandruff, bacteriostatic and deodorant products.
Some biosurfactants are used in the pharmaceutical and biomedical industry for their antimicrobial and / or antifungal activity..
In the food industry
In the food industry, biosurfactants are used in the manufacture of mayonnaise (which is an emulsion of egg water and oil). These biosurfactants come from lectins and their derivatives, which improve quality and additionally taste..
In the agriculture
In agriculture, biosurfactants are used for the biological control of pathogens (fungi, bacteria, viruses) in crops.
Another use of biosurfactants in agriculture is to increase the availability of micronutrients from the soil..
References
- Banat, I.M., Makkar, R.S. and Cameotra, S.S. (2000). Potential commercial applications of microbial surfactants. Applied Microbiology Technology. 53 (5): 495-508.
- Cameotra, S.S. and Makkar, R.S. (2004). Recent applications of biosurfactants as biological and immunological molecules. Current Opinions in Microbiology. 7 (3): 262-266.
- Chen, S.Y., Wei, Y.H. and Chang, J.S. (2007). Repeated pH-stat fed-batch fermentation for rhamnolipid production with indigenous Pseudomonas aeruginosa Applied Microbiology Biotechnology. 76 (1): 67-74.
- Mulligan, C.N. (2005). Environmental applications for biosurfactants. Environmental Pollution. 133 (2): 183-198.doi: 10.1016 / j.env.pol.2004.06.009
- Tang, J., He, J., Xin, X., Hu, H. and Liu, T. (2018). Biosurfactants enhanced heavy metals removal from sludge in the electrokinetical treatment. Chemical Engineering Journal. 334 (15): 2579-2592. doi: 10.1016 / j.cej.2017.12.010.



Yet No Comments