
Spore staining rationale, techniques and uses

The spore staining It is the methodology used to color the resistance structures that form some bacterial genera when they are in unfavorable conditions; these structures correspond to a form of survival.
There are many genera that form spores; however, the main ones are Bacillus and Clostridium. These genera are considered more relevant because they have species that are pathogenic for humans..
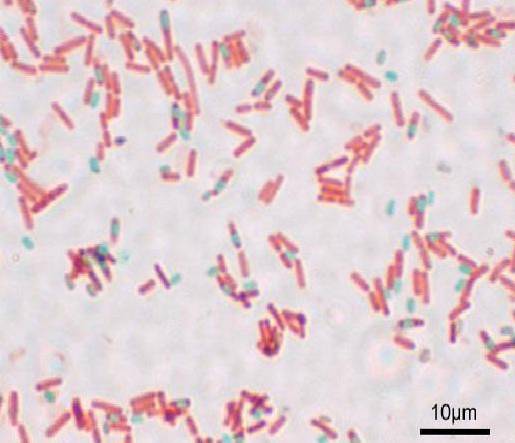
Each bacillus can give rise to a spore. At the time of staining the preparation, the spore can be found within the bacillus (endospore) or outside it (exospore). With conventional staining techniques for bacteria -such as the Gram stain- the spores are colorless.
Currently, there are several staining methodologies that are capable of penetrating the thick structure of the spore to dye it. These methodologies are very varied; These include the Dorner technique, the Möeller stain and the Shaeffer-Fulton methodology, also known as Wirtz-Conklin..
Of all the techniques mentioned, the Shaeffer-Fulton methodology is the most widely used in routine laboratories. It is named after two microbiologists who created the coloration in 1930: Alicia Shaeffer and MacDonald Fulton. However, the technique is sometimes called Wirtz-Conklin in honor of two bacteriologists from the 1900s..
Article index
- 1 Rationale
- 2 Spore staining techniques
- 2.1 Dorner technique
- 2.2 Modified Dorner Technique
- 2.3 Shaeffer-Fulton or Wirtz-Conklin technique
- 2.4 Möeller technique
- 2.5 Modified Möeller technique without heat
- 3 Uses
- 3.1 Examples
- 4 References
Basis
The spores do not stain with conventional stains because they have a very thick wall. The complex composition of the spores prevents the entry of most dyes.
If the spore is studied from the outside in, the following layers are observed: first is the exosporium, which is the thinnest and outer layer formed by glycoproteins.
Next comes the cuticle, which provides resistance to high temperatures, followed by the cortex composed of peptidoglycan. Later is the wall of the base that protects the protoplast.
The spore is a dehydrated structure that contains 15% calcium and dipicolinic acid. For this reason, most spore staining techniques are based on the application of heat so that the dye can penetrate the thick structure..
Once the spore is stained, it cannot remove the dye. In the Shaeffer-Fulton technique, malachite green enters vegetative cells and, when heat is applied, penetrates the endospore and also the exospores.
By washing with water, the dye is removed from the vegetative cell. This occurs because the malachite green dye is slightly basic, so it binds weakly to the vegetative cell..
Instead, it cannot get out of the spore and the bacillus is eventually counterstained with safranin. This foundation is valid for the rest of the techniques, in which something similar happens.
Spore staining techniques
To perform the spore staining, you must have a pure culture of the suspicious strain you want to study..
The culture is subjected to extreme temperatures for 24 hours to stimulate the microorganism to sporulate. For this, the culture can be placed in an oven at 44 ° C or in a refrigerator (8 ° C) for 24 or 48 hours..
If left too long at the mentioned temperatures, only exospores will be observed, since all endospores will have already left the bacillus.
At the end of the time, a few drops of sterile physiological solution should be placed on a clean slide. Then a small portion of the culture is taken and a fine spread is made.
Later it is left to dry, it is fixed to the heat and it is dyed with one of the techniques that are explained below:
Dorner Technique
1- Prepare a concentrated suspension of the sporulated microorganism in distilled water in a test tube and add an equal volume of filtered Kinyoun carbol fuchsin.
2- Place the tube in a boiling water bath for 5 to 10 minutes.
3- On a clean slide, mix a drop of the previous suspension with a drop of 10% nigrosine aqueous solution, boiled and filtered..
4- Spread and dry quickly with gentle heat.
5- Examine with a 100X objective (immersion).
Spores stain red and bacterial cells appear almost colorless against a dark gray background.
Modified Dorner Technique
1- A suspension of the sporulated microorganism is made on a slide and fixed in the heat.
2- The sample is covered with a filter paper strip to which carbolic fuchsin is added. The colorant is heated for 5 to 7 minutes with the flame of the Bunsen burner until the evolution of vapors is generated. Then the paper is removed.
3- The preparation is washed with water and then dried with absorbent paper.
4- Cover the smear with a thin film of 10% nigrosin, using a second slide to spread the nigrosin or a needle.
The coloration that spores and bacteria take is the same as that described in the prior art.
Shaeffer-Fulton or Wirtz-Conklin technique
1- Make a fine smear with a suspension of the sporulated microorganism on a slide and heat fix.
2- Cover the slide with 5% malachite green aqueous solution (a filter paper can be placed on the slide).
3- Heat over the Bunsen burner flame to cause the release of vapors and remove the flame. Repeat the operation for 6 to 10 minutes. If the malachite green solution evaporates too much during the procedure, more.
4- Remove the filter paper (if installed) and wash with water.
5- Cover the slide with 0.5% aqueous safranin for 30 seconds (some variants of the technique use 0.1% aqueous safranin and leave it for 3 minutes).
With this technique the spores appear green and the bacilli red..
It has the disadvantage that the endospores of young cultures do not stain well, since they appear extremely clear or colorless. To avoid this, it is recommended to use cultures of 48 hours of incubation..
Möeller technique
1- Cover the smear with chloroform for 2 minutes.
2- Discard the chloroform.
3- Cover with 5% chromic acid for 5 minutes.
4- Wash with distilled water
5- The sheet is covered with carbol fuchsin-fenicada and it is exposed to the flame of the Bunsen burner until the emission of vapors; then it is removed from the flame for a few moments. The operation is repeated until 10 minutes are completed.
6- Wash with water.
7- Use acidified ethanol (hydrochloric alcohol) to discolor. Leave for 20 or 30 seconds.
8- Wash with distilled water.
9- Contrastinate by covering the sheet with methylene blue for 5 minutes.
10- Wash with distilled water.
11- Let it dry and take the sample to the microscope.
The spores appear red and the bacilli blue. It is important not to breathe in the vapors, as they are toxic and in the long term can be carcinogenic..
Heatless Modified Möeller Technique
In 2007 Hayama and his collaborators created a modification of the Möeller technique. They eliminated the step of heating the dye and replaced it with the addition of 2 drops of the surfactant Tergitol 7 for every 10 ml of carbol fuchsin-carbol solution. The same results were obtained.
Applications
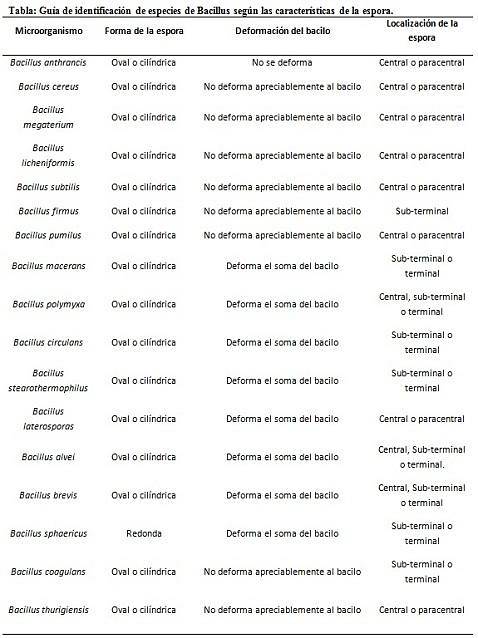
The staining of spores provides very valuable and useful information for the identification of the pathogen, since its presence, its shape, location within the bacillus and the ability to deform the vegetative cell or not, are data that can guide the species. involved within a certain gender.
In this context, it is worth saying that the spores can be round or oval, they can be located in the center or also in a paracentral, subminal or terminal position..
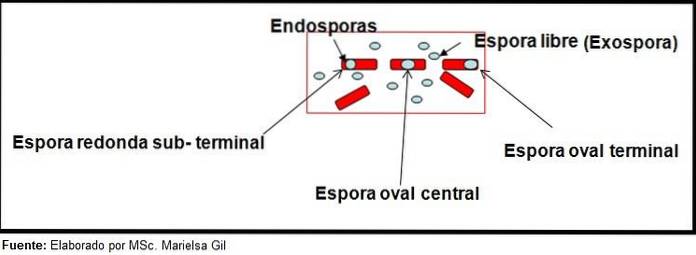
Examples
- Clostridium difficile forms an oval spore in terminal position that deforms the bacillus.
- The spore of Clostridium tertium it is oval, does not deform the bacillus and is located at the terminal level.
- The endospore of Clostridium tetani is terminal and deforms the bacillus, giving the appearance of a drumstick.
- Spores of Clostridium botulinum, C. histolyticum, C. novy Y C. septicum are round or oval subterminal and deform the bacillus.
- The endospore of Clostridium sordelli is located in the central position, with a slight deformation.
References
- Hayama M, Oana K, Kozakai T, Umeda S, Fujimoto J, Ota H, Kawakami Y. Proposal of a simplified technique for staining bacterial spores without applying heat-successful modification of Moeller's method. Eur J Med Res. 2007; 16 12 (8): 356-9.
- Wikipedia contributors. Moeller stain. Wikipedia, The Free Encyclopedia. November 3, 2018, 03:28 UTC. Available at: en.wikipedia.org
- Pérez R, Juárez M, Rodríguez (2011). Microbiological Techniques Laboratory Manual. Department of Basic Sciences Academy of Microbiology. National Polytechnic Institute.
- "Endospore." Wikipedia, The Free Encyclopedia. 25 Feb 2018, 10:20 UTC. 10 Jan 2019, 02:42: en.wikipedia.org
- Silva L, Silva C, Fernández N, Bueno C, Torres J, Rico M, Macías J and collaborators. (2006). Labor Personnel of the autonomous community of Extremadura. Specific agenda Volume IV. Editorial MAD. Seville-Spain, pp 211-212.
- Silva M, García M, Corrales J, Ponce E. (2006). Laboratory specialist technician, Galician Health Service (SERGAS). Specific subject agenda volume 2. Editorial MAD. Seville-Spain, pp 79-80.
- Koneman E, Allen S, Janda W, Schreckenberger P, Winn W. (2004). Microbiological Diagnosis. (5th ed.). Argentina, Editorial Panamericana S.A.
- Forbes B, Sahm D, Weissfeld A. 2009. Bailey & Scott Microbiological Diagnosis. 12 ed. Argentina. Editorial Panamericana S.A



Yet No Comments