
May Grünwald-Giemsa stain rationale, technique and uses

The May Grünwald-Giemsa stain o Pappenheim is a differential staining technique that mixes Giemsa and May Grünwald reagents. It is used for the differentiation of normal and abnormal blood cells in peripheral blood and bone marrow smears, as well as for the staining of histological sections and cytological samples..
Both reagents -Giemsa and May Grünwald- are derived from Romanowsky-type staining, a technique that is based on the combination of acidic and basic dyes..
Giemsa improved the technique by stabilizing the mixture of eosin, methylene blue and their derivatives, with glycerol. Instead, May Grünwald uses eosin and methylene blue, using methanol as the solvent. This strategic combination has yielded excellent results.
Although in terms of the observation of cell morphology, it acts in a similar way to the Giemsa and Wright stains, this technique improves the previous ones by refining the staining of the parasites that cause malaria, Chagas disease, leishmaniasis and trichomoniasis..
Additionally, it has proven to be a very useful technique for the cytological study of spermatic fluid. It has stood out not only by showing the morphological characteristics of spermatozoa, but also by allowing the differentiation of leukocytes, epithelial cells and spermatogenesis cells with great efficiency..
Article index
- 1 Rationale
- 1.1 Variety of colorants
- 2 Technique
- 2.1 Materials
- 2.2 Concentrated solution of May Grünwald dye
- 2.3 Concentrated Giemsa stain
- 2.4 Preparation of buffer solution at pH 7.2
- 2.5 Blood or bone marrow smear staining procedure
- 2.6 Technique for coloring smears of spermatic fluid
- 2.7 Important specifications
- 3 Uses
- 3.1 Vaginal cytology
- 3.2 Sperm sample
- 4 References
Basis
The technique follows the foundation of Romanowsky stains, in which acidic dyes have selective affinity for basic cellular components and acidic components attract basic stains..
Explained in another way, both cell structures and dyes have positive or negative electrical charges; like charges repel and different charges attract.
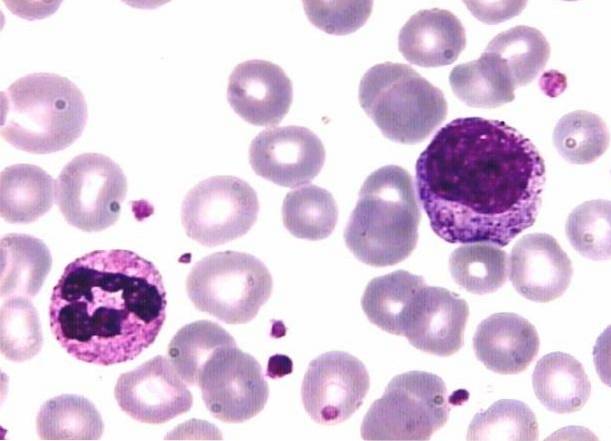
For example, basic dyes like methylene blue are positively charged and attracted to negatively charged structures. That is why this dye stains nuclei that are rich in DNA and RNA that have negatively charged phosphate groups..
The granules of segmented basophils and the cytoplasms of mononuclear white blood cells containing RNA are also stained..
Likewise, the acid dye carries a negative charge, which is why it binds to positively charged structures such as erythrocytes and the granules of segmented eosinophils. As for the granules of the segmented neutrophils, these fix both dyes.
Variety of colorants
In this technique there is a combination of reactions between orthochromatic and metachromatic dyes. Orthochromatics (eosin and methylene blue) bind to the cell structure to which they are related and provide a stable color that does not vary.
On the other hand, the metachromatics (the derivatives of methylene blue azure A and azure B), vary their original color once attached to the specific structure, and there may even be a variety of shades.
Finally, the step that the May Grünwald solution takes requires the presence of water, since without this the dye will penetrate the structures but will not set. For this to happen, the dye must become polar or ionize, and thus be able to precipitate and bind to related structures..
Technique
Materials
- Microscope slides.
- Bridges of coloring.
- May-Grünwald solution.
- Giemsa stain.
- Distilled water.
May Grünwald dye concentrated solution
0.25 g of eosin-methylene blue (stain according to May Grünwald) should be weighed out and dissolved in 100 ml of methanol. Then the preparation is mixed for 1 hour and left to rest for 24 hours. When the time is up, it leaks.
To apply the technique, the May Grünwald dye must be diluted as follows: for 200 ml of diluted dye, measure 30 ml of the concentrated solution, add 20 ml of buffer solution and 150 ml of distilled water adjusted to pH7.2-7.3 . Later it is mixed and filtered.
Giemsa stain concentrate
0.5 g of azure-eosin-methylene blue (stain according to Giemsa) must be weighed, dissolved in 50 ml of methanol and 50 ml of glycerin added to the mixture.
To perform the technique, it is diluted 1:10 with buffer solution and allowed to stand for 10 minutes. Can be filtered if necessary.
Preparation of the buffer solution at pH 7.2
They should be weighed:
- 40 mg potassium di-hydrogen phosphate (KH2PO4).
- 151 mg di-sodium hydrogen phosphate 12-hydrate (Na2HPO4).
Both compounds are dissolved in 100 ml of water.
Blood or bone marrow smear staining procedure
There are two modes: a classic and a fast.
Classic mode
- Cover the smears for 2 to 3 minutes with the diluted May-Grünwald solution..
- Wash with buffered distilled water to remove the previous solution.
- Cover with the same buffered wash solution and leave for 1 minute. The idea is that the previous dye is fixed to the structures and that, at the same time, the cells are hydrated.
- Add 12 drops of diluted Giemsa tincture to the buffered water and blow to mix and homogenize. Let stand for 15-20 minutes.
- Wash smears with buffered distilled water and place to air dry.
- Focus and observe the stained blood cells under a light microscope using the 40X objective. If necessary, the 100X can be used.
Quick mode
- Cover the smear with diluted May Grünwald stain for 1 minute..
- Wash with buffered distilled water.
- Cover with buffered water and let stand for 1 minute.
- Place the diluted Giemsa stain and leave for 5 minutes.
- Wash with buffered distilled water and allow to air dry.
The techniques described here are a guideline, but it should be taken into account that the procedures and staining times vary according to the commercial company that distributes the reagents. It is advisable to follow the steps strictly indicated by each commercial house.
Technique for coloring smears of spermatic fluid
1- Cover the smear with a thin layer of the May Grünwald solution for 4 minutes.
2- Remove the dye and wash with distilled water.
3- Place a layer of diluted Giemsa (1:10) in distilled water for 15 minutes.
4- Remove the dye and wash with distilled water.
5- Let dry and observe in the microscope.
Important Specifications
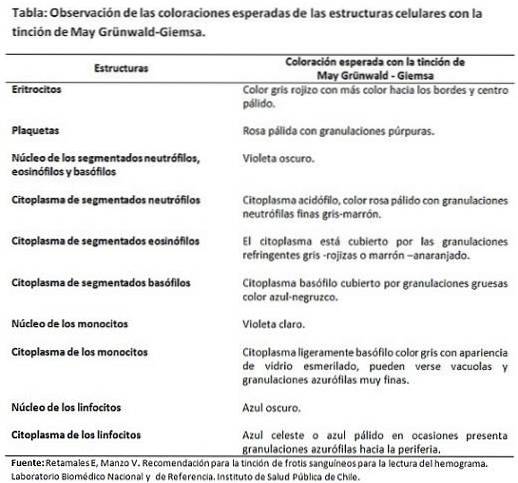
The technique requires that the reagents and washing solutions have a pH adjusted to 7.2 -7.3, so that the affinities of the dyes for the cell structures are not distorted and the expected final color does not vary..
Applications
This technique is used by clinical laboratories to stain peripheral blood and bone marrow smears, tissue sections, and cytologies..
In the hematological field, this technique is of vital importance in the study of cell abnormalities in terms of shape, size and number. It is a very valuable tool for the diagnosis of certain diseases, such as leukemias and anemias.
In addition, it is of outstanding utility when looking for parasites in hematological settings (Plasmodium sp Y Trypanosome cruzi) or histological (Leishmanias sp).
Vaginal cytology
Regarding vaginal cytology, this technique is especially advantageous for the observation of Trichomonas vaginalis. This is an important finding, since its presence simulates pictures of carcinoma. in situ which then disappear when the parasite is removed.
Sperm sample
It has been an ideal tool for the study of sperm samples, since it provides valuable information on the quality of the sperm.
The data it offers has to do mainly with number and morphology, as well as with the concomitant cells that may be present and that are of vital importance, such as germ cells, leukocytes and epithelial cells..
With this analysis it is possible to describe abnormalities observed in the spermatozoa in the head, neck, midpiece and main part..
In addition, they can also help to show cases of hemospermia (presence of red blood cells in semen) and leucospermia or piospermia (increased number of leukocytes in semen).
References
- Costamagna S, Prado M. Validation of the fresh test, May Grünwald-Giemsa and Gram stains and culture media for the diagnosis of Trichomonas vaginalis. Parasitol. 2001; 25 (1-2): 60-64. Available in: scielo.
- Merck KGaA Laboratory. May Grünwald eosin methylene blue for microscopy.
- "May-Grünwald-Giemsa stain." Wikipedia, The Free Encyclopedia. 15 Nov 2018, 14:37 UTC. 8 Jan 2019, 04:29: en.wikipedia.org
- Glass Chemicals Panreac Laboratory. Reagents for histological techniques, hematology and microbiology. Available at: glasschemicals.com
- Retamales E, Manzo V. Recommendation for staining of blood smears for reading the hemogram. National and Reference Biomedical Laboratory. Institute of Public Health of Chile.
- Sarabia L. Spermiogram according to WHO criteria. Developmental Anatomy and Biology Program. School of Medicine. University of Chile. Available at: pp.centramerica.com



Yet No Comments