
Tubulin Alpha and Beta, Functions
The tubulin is a globular dimeric protein made up of two polypeptides: alpha and beta tubulin. They are organized in the shape of a tube to give rise to microtubules, which together with actin microfilaments and intermediate filaments make up the cytoskeleton.
Microtubules are found in different essential biological structures, such as the flagella of sperm, the extensions of ciliated organisms, the cilia of the trachea and fallopian tubes, among others..
In addition, the structures that tubulin forms function as transport routes - analogous to the tracks of a train - of materials and organelles within the cell. The movement of substances and structures is possible thanks to motor proteins associated with microtubules, called kinesin and dynein..
Article index
- 1 General characteristics
- 2 Alpha and beta tubulin
- 3 Functions
- 3.1 Cytoskeleton
- 3.2 Mitosis
- 3.3 Centrosome
- 4 Evolutionary perspective
- 5 References
General characteristics
Tubulin subunits are 55,000 dalton heterodimers and are the building blocks of microtubules. Tubulin is found in all eukaryotic organisms and has been highly conserved throughout evolution.
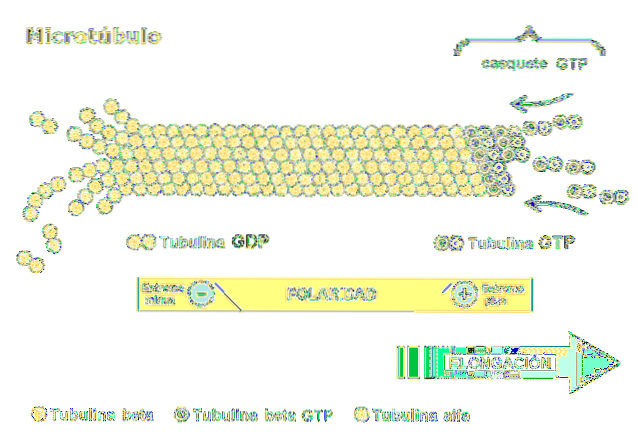
The dimer is made up of two polypeptides called alpha and beta tubulin. These polymerize to form microtubules, which consist of thirteen protofilaments arranged in parallel in the shape of a hollow tube..
One of the most relevant characteristics of microtubules is the polarity of the structure. In other words, the two ends of the microtubule are not the same: one end is called the fast-growing or "more" end, and the other is the slow-growing or "less" end..
Polarity is important as it determines the direction of movement along the microtubule. Tubulin dimer is capable of polymerizing and depolarizing in rapid assembly cycles. This phenomenon also occurs in actin filaments.
There is a third type of subunit: it is gamma tubulin. This is not part of the microtubules and is located in the centrosomes; however, it participates in the nucleation and formation of microtubules.
Alpha and beta tubulin
The alpha and beta subunits associate strongly to form a complex heterodimer. In fact, the interaction of the complex is so intense that it does not dissociate under normal conditions..
These proteins are made up of 550 amino acids, mostly acidic. Although alpha and beta tubulins are quite similar, they are encoded by different genes..
In alpha tubulin, amino acid residues with an acetyl group can be found, giving it different properties in cell flagella..
Each subunit of tubulin is associated with two molecules: in alpha tubulin, GTP binds irreversibly and hydrolysis of the compound does not occur, while the second binding site in beta tubulin, reversibly binds GTP and hydrolyzes it..
GTP hydrolysis results in a phenomenon called "dynamic instability" where microtubules undergo growth and decline cycles, depending on the rate of tubulin addition and the rate of GTP hydrolysis..
This phenomenon results in a high turnover rate of the microtubules, where the half-life of the structure is only a few minutes..
Features
Cytoskeleton
The alpha and beta subunits of tubulin polymerize to give rise to microtubules, which are part of the cytoskeleton.
In addition to microtubules, the cytoskeleton is made up of two additional structural elements: actin microfilaments of approximately 7 nm and intermediate filaments of 10 to 15 nm in diameter..
The cytoskeleton is the framework of the cell, it supports and maintains the cell shape. However, the membrane and subcellular compartments are not static and are in constant movements to be able to carry out the phenomena of endocytosis, phagocytosis and secretion of materials..
The structure of the cytoskeleton allows the cell to accommodate itself to fulfill all the aforementioned functions..
It is the ideal medium for cell organelles, the plasma membrane and other cell components to perform their usual functions, in addition to participating in cell division.
They also contribute to cellular movement phenomena such as amoeba locomotion, and specialized structures for movement such as cilia and flagella. Finally, it is responsible for the movement of the muscles.
Mitosis
Thanks to dynamic instability, microtubules can be completely reorganized during cell division processes. The microtubule arrangement during the interface is capable of disassembling and the tubulin subunits are free.
Tubulin can reassemble and form the mitotic spindle, which is involved in the separation of chromosomes.
There are certain drugs, such as colchicine, taxol, and vinblastine that disrupt the processes of cell division. It acts directly on tubulin molecules, affecting the assembly and dissociation phenomenon of microtubules.
Centrosome
In animal cells, microtubules extend into the centrosome, a structure close to the nucleus made up of a pair of centrioles (each oriented perpendicularly) and surrounded by an amorphous substance, called the pericentriolar matrix..
The centrioles are cylindrical bodies formed by nine triplets of microtubules, in an organization similar to the cilia and cell flagella.
In the process of cell division, the microtubules extend from the centrosomes, thus forming the mitotic spindle, responsible for the correct distribution of the chromosomes to the new daughter cells..
It seems that centrioles are not essential for the assembly of microtubules within cells, since they are not present in plant cells or in some eukaryotic cells, such as in the ovules of certain rodents..
In the pericentriolar matrix, the initiation for the assembly of microtubules occurs, where nucleation occurs with the help of gamma tubulin.
Evolutionary perspective
The three types of tubulin (alpha, beta, and gamma) are encoded by different genes and are homologous to a gene found in prokaryotes that encodes a 40,000-dalton protein, called FtsZ. Bacterial protein is functionally and structurally similar to tubulin.
It is likely that the protein had an ancestral function in bacteria and was modified during evolutionary processes, concluding in a protein with the functions it performs in eukaryotes.
References
- Cardinali, D. P. (2007). Applied neuroscience: its fundamentals. Panamerican Medical Ed..
- Cooper, G.M. (2000). The Cell: A Molecular Approach. 2nd edition. Sunderland (MA): Sinauer Associates.
- Curtis, H., & Schnek, A. (2006). Invitation to Biology. Panamerican Medical Ed..
- Frixione, E., & Meza, I. (2017). Living Machines: How Cells Move?. Fund of Economic Culture.
- Lodish H, Berk A, Zipursky SL, et al. (2000). Molecular Cell Biology. 4th edition. New York: W. H. Freeman.



Yet No Comments