
30 Examples of Chemical Suspensions

Suspensions are heterogeneous mixtures that are studied in chemistry. Heterogeneous mixtures are those in which the particles that make them up can be distinguished.
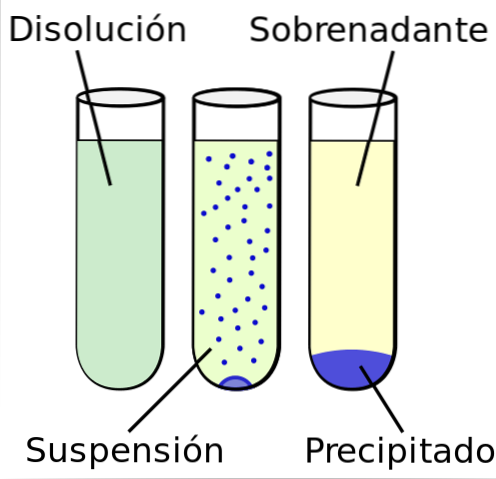
These are formed by one or more substances that are in solid state suspended in a liquid medium. To be a suspension and not a solution, solid particles cannot be soluble in the liquid medium.
The particles in the suspension must be greater than one micron in size. Some solutions prevent the light from passing through correctly, even becoming opaque.
Suspensions can be separated into solid and liquid particles through decantation, filtration, centrifugation or evaporation.
Once the suspension is made, some of the particles can be added to others, so if we want to maintain the suspension, surfactants or dispersing agents are usually added to the liquid medium..
Suspensions, solutions and colloids
A distinction must be made between suspensions, solutions and colloids. Solutions are homogeneous mixtures, where solid particles are dispersed in the liquid medium, changing the atomic or ionic level. Colloids are heterogeneous mixtures where solid particles are less than one micron in size..
Four phases can be distinguished in a suspension. The first phase is the solid phase or internal phase where the solid particles cannot be divided in the suspension..
In the external phase, or also known as the liquid phase, it is where the solid particles are at rest in the liquid part.
In the surfactant part of the suspension, the elements do not come together or aggregate. And finally, we can use stabilizers in the suspension to increase its strength and prevent the particles from deteriorating. These stabilizers can be thickeners, antifreezes or preservatives.
Examples of suspensions
1-Fruit juice: these are suspensions since the pulp of the fruits floats in the liquid medium. If we only want to have the liquid medium, we will have to decant or filter the mixture.
2-Cloudy river water: in this suspension the sediments that carry the river form the suspension.
3-Watercolors: they are a suspension that is deposited on the paper where it filters the water and collects the color pigment
4-Powdered drugs: to keep them in suspension and not settle to the bottom, they have to be stirred.
5-Exfoliating creams: where there are small particles forming solid grains in the cream to fulfill the exfoliation function.
6-Milk: animal fat particles are found in solution with water. As they are less dense than the dispersant, they tend to stay on the surface over time
7-Paint: it is a suspension of color pigments in an aqueous or oily medium. If it is not agitated, it can become separated.
8-Sea water: In the shore area it can be considered a suspension with sand particles, although this suspension is of limited duration.
9-Dressings for salads: they contain vegetable particles suspended in oil or vinegar, they have a viscous dispersant that keeps them in a state of rest.
10-Injectable drug suspensions: drugs are found in solution in a saline so that they can access the bloodstream more easily.
Other examples of common suspensions
11-Horchata water
12-Cocoa in milk or water
13-Moisturizing creams or facial creams
14-Penicillin
15-Insulin
16-Amoxicillin (antibiotic)
17-Powder makeup
18-Ash in a volcanic eruption
19-The whitewash
20-Powdered milk
Examples of pharmaceutical suspensions
Pharmaceutical suspensions are used when the drug is insoluble on its own, in addition to being more stable in suspension or emulsion form. Once the drugs need to be controlled, when used in a suspension, the release rate of the active ingredient can be controlled..
And one of the biggest reasons to use suspension and injectable drugs is because patients do not tolerate the bad taste of drugs or their pill form..
21-Antacid suspensions (used as a medicine for heartburn) are suspensions of magnesium hydroxide or aluminum hydroxide. Medicines like Mylanta or AciTip
22-Corticosteroid medication suspensions. Examples can be such as Diprospan, Scherin
23-Suspension of white clay (kaolin) as antidiarrheal methods
24-Suspensions of antiparasitic drugs. For example, metronidazole suspensions
25-Suspensions for oral use, these suspensions are prepared mainly so that they do not need to be injected, but can be taken orally.
26-Otic suspensions, where the suspensions are prepared to be used inside the ears in a superficial way.
27-Topical suspensions: they are prepared to be used directly on the skin without the need to be injected
28-Ophthalmic suspensions: they are a suspension with a specific neutral pH to use in the eyes
29-Injectable suspensions: these are the most widespread suspensions within the field of pharmaceuticals, where the drug is in suspension to be used intravenously through an injection.
30-Rectal suspensions: these are prepared to be used rectally as a suppository, usually through an enema.
References
- Remington "The Science and Remington" The Science and Practice of Pharmacy "20 Practice of Pharmacy" 20 th Edition, United Stat Edition, United States of America (2000)
- Remington Remington, Pharmacy, Volume I, 19th, Pharmacy, Volume I, 19th ed. Editorial Médica Panamerica Editorial Médica Panamericana, Buenos Aires; 1998.
- Vila Jato, Vila Jato, J.L ,. "Pharmaceutical Technology", Vol. I and II. "Pharmaceutical Technology", Vol. I and II, Ed. Synthesis, Madrid (1997)
- COTTON, F. AlbertWilkinson, et al. Basic inorganic chemistry. Limusa ,, 1996.
- HIMMELBLAU, David M. Basic principles and calculations in chemical engineering. Pearson Education, 1997.
- SKOOG, Douglas Arvid, et al. Analytic chemistry. McGraw-Hill Interamericana ,, 1995.
- VIAN, Angel; OCÓN, Joaquín. Chemical engineering elements: (basic operations). Aguilar, 1957.



Yet No Comments