
Acinetobacter baumannii characteristics, morphology, symptoms
Acinetobacter baumannii It is a gram-negative bacterium, belonging to the order of Pseudomonas. Throughout its taxonomic history it has been located in the genera Micrococcus, Moraxella, Alcaligenes, Mirococcuscalco-aceticus, Herellea Y Achromobacter, until it was placed in its current genre in 1968.
TO. baumannii it is a pathogenic bacterium, considered the species most frequently implicated in infections within its genus. It has been identified as being involved in various types of infections such as septicemia, pneumonia and meningitis.
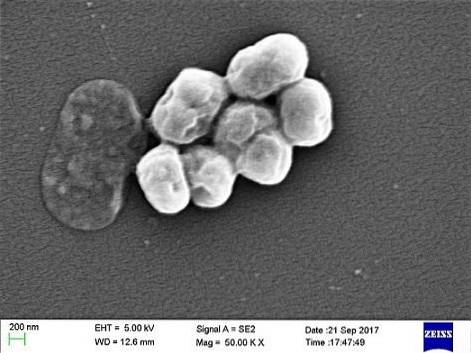
It is an opportunistic parasite that has a significant incidence in hospital or nosocomial diseases. Transmission by mechanical ventilation has been indicated as an important cause of contagion, especially in Intensive Care Units.
Despite being considered a low-grade pathogen, it has the ability to increase its virulence in strains involved in frequent infections. It has a great capacity to develop resistance and multi-resistance to antibiotics.
In hospitals it is transmitted mainly by the contact of staff, by the use of contaminated hospital materials and also through the air over short distances.
The World Health Organization includes A. baumannii on a list of resistant pathogens for which new antibiotics are urgently required, assigning it category 1, with critical priority.
Article index
- 1 Characteristics of Acinetobacter baumannii
- 2 Morphology
- 3 Metabolism
- 4 Resistance to antibiotics
- 5 Pathologies
- 6 Symptoms
- 7 Treatment
- 8 References
Characteristics of Acinetobacter baumannii
All species of the genus Acinetobacter have a wide distribution in different natural niches. A. baumannii it can naturally inhabit the skin of healthy people, being able to colonize mucosal surfaces, which constitutes an important epidemiological reservoir. However, the habitat of A. baumannii is almost exclusive to hospital settings.
These bacteria lack flagella or structures used for locomotion. However, they achieve movement through structures that allow them to extend and retract, and through chemical mechanisms such as the excretion of a film of a high molecular weight exopolysaccharide behind the bacteria..
A. baumannii can colonize a large number of living or inert environments and has a great ability to survive on artificial surfaces for an extended period of time.
This ability is possibly due to their ability to resist dehydration, to use various carbon sources through various metabolic pathways, and because of the ability to form biofilms. For this reason it is common to find it in hospital material, such as catheters and mechanical ventilation devices..
Morphology
A. baumannii is a coccobacillus, with an intermediate shape between cocci and rods. They measure 1.5 to 2.5 by 1 to 1.5 microns when populations are in a logarithmic phase of growth. They are more spherical when they reach the stationary phase.
Metabolism
The bacteria A. baumannii it is not a glucose fermenter; it is strict aerobic, that is, it requires oxygen for its metabolism.
The species of the genus Acinetobacter they are the only ones of the Moraxellaceae family that lack cytochrome c oxidases, for which they give negative results in oxidase tests.
A. baumannii grows at temperatures that vary between 20 and 44ºC, being its optimum temperature between 30 and 35ºC.
Antibiotic resistance
The constant generation of resistance to antibiotics not only hinders the treatment and control of infections caused by A. baumannii, it also promotes the selection of endemic and epidemic multi-resistant strains.
Some intrinsic mechanisms of A. baumannii, that promote antibiotic resistance:
- The presence of β-lactamases confers resistance to b-lactams.
- Production of specific enzymes such as ammoniglucoside-3'-phosphotransferase VI inactivates amikacin.
- The presence of oxacillinase OXA-51 hydrolyzes penicillins and carbapenems.
- The presence and overexpression of reflux pumps, which are pumps that expel small molecules out of the cell that manage to penetrate the cytoplasm, thus reducing their susceptibility to antibiotics.
The biofilms generated by A. baumannii alter the metabolism of microorganisms, reducing their sensitivity to antibiotics, in addition to providing a physical barrier against large molecules and preventing dehydration of bacteria.
Pathologies
A. baumannii colonizes a new host by contact with infected people, or with contaminated medical equipment. First of all, this bacterium attaches itself to the skin and mucosal surfaces. To reproduce, it must survive antibiotics and inhibitors and the conditions of these surfaces..
The increase in the number of bacteria on mucosal surfaces, particularly in hospitalization conditions in contact with intravascular catheters or endotracheal tubes, may increase the risk of infection of the respiratory tract and bloodstream..
Hospital-acquired pneumonia is the most common of the infections caused by A. baumanii. Commonly contracted in Intensive Care Units, by patients receiving mechanically assisted respiration.
A. baumanii it has also caused major infection problems in military personnel with postwar trauma, specifically in Iraq and Afghanistan. Specifically, due to osteomyelitis and soft tissue infections, which can cause necrosis and cellulitis.
There are also risks of meningitis from A. baumanii in patients recovering from neurosurgery.
Individuals susceptible to infections by A. baumanii include those who have previously been subjected to the use of antibiotics, major surgeries, burns, trauma, immunosuppression or the use of invasive medical devices, mainly mechanical ventilation, in Intensive Care Units.
Symptoms
There is no specific symptomatology of infections by A. baumanii. Each of the different infections produced by this bacterium has its own characteristic symptoms..
In general the symptoms of infections that may involve A. baumanii or other opportunistic bacteria such as Klebsiella pneumoniae Y Streptococcus pneumoniae, may include fever, chills, rash, painful urination, urgent need to urinate frequently, confusion or altered mental states, nausea, muscle pain, chest pain, and cough.
Treatment
Antibiotics to treat infections by A. baumannii they are extremely limited, due to their great ability to acquire resistors and multi-resistors. For this reason, it is important to determine the susceptibility of each strain to different antibiotics to guarantee the effectiveness of each treatment..
Given resistance to carbapenems, polymyxins, specifically colistin, have been used, despite having a relatively low resistance index and their side effects on the kidneys.
However, colistin resistant strains have already been detected. As an alternative to resistance to these antibiotics, combination therapy has been used.
References
- Bergogne-Bérézin, E. & Towner, K.J. Acinetobacter spp. as nosocomial pathogens: microbiological, clinical and epidemiological features. Clin Microbiol Rev, 9 (1996), pp. 148-165.
- Fournier, P.E., Richet, H. (2006). The Epidemiology and Control of Acinetobacter baumanii in Health Care Facilities. Clinical Infectious Diseases, 42: 692-9.
- Hernández Torres, A., García Vázquez, E., Yagüe, G. &, Gómez Gómez, J. (2010) Acinetobacter baumanii Multiresistant: current clinical situation and new perspectives Revista Española de Quimioterapia, 23 (1): 12-19.
- Maragakis LL, Perl TM. Acinetobacter baumanii: Epidemiology, Antimicrobial Resistance and Treatment Options. Clin Infec Dis 2008; 46: 1254-63.
- McConnell, M.j., Actis, L. & Pachón, J. (2013) Acinetobacter baumannii: human infections, factors contributing to pathogenesis and animal models. FEMS Microbiology Reviews, 37: 130-155.
- Peleg, A.Y., Seifert, H. & Paterson, D.L. (2008). Acinetobacter baumannii: emergence of a successful pathogen. Clinical Microbiology Reviews, 21 (3): 538-82.
- Vanegas-Múnera, J.M., Roncancio-Villamil, G. & Jiménez-Quiceno, J.N. (2014). Acinetobacter baumannii: clinical importance, resistance mechanisms and diagnosis. CES Medicine Magazine, 28 (2): 233-246.

Yet No Comments