
Biuret rationale, reagents, procedure, uses

The Biuret is a reagent used for the determination of long chain and short chain proteins. It is especially used in the area of analytical chemistry and urinalysis to investigate the concentration of total proteins in serum, plasma and urine..
Protein values can be increased or decreased in certain pathologies. Hypoproteinemia symptoms usually occur in patients with kidney disease, in malnourished patients, and in patients with chronic infections..
While hyperproteinemia is observed in pathologies such as multiple myeloma, systemic lupus erythematosus, bacterial endocarditis, bacterial meningitis, Waldenstrom's macroglobulinemia, among others..
On the other hand, the presence of protein in the urine is due to the filtration of albumin by the kidney. This is a pathological behavior that should be studied.
In this sense, Biuret is very useful, as it allows quantifying the presence of proteins in serum, plasma, urine, among many other samples.
Biuret can even be used to investigate the presence and concentration of proteins in little-explored samples or samples of unknown composition. Therefore, it is widely used in the research area.
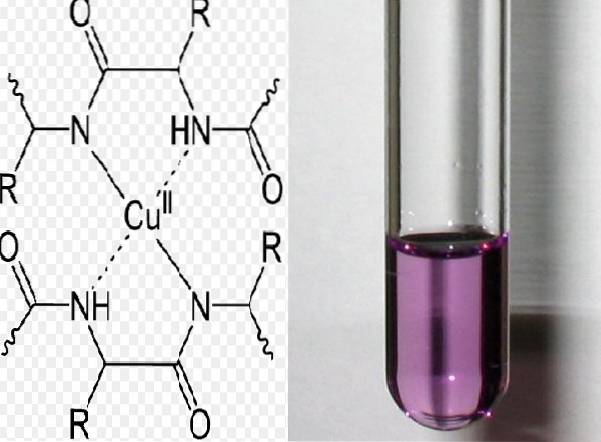
The Biuret test is based on detecting peptide bonds. The test takes place in an alkaline medium. The sample must contain at least two peptide bonds for a violet-purple complex to form. The complex is formed by the union of the bonds and the copper ion.
Article index
- 1 Rationale
- 2 Reagents
- 2.1 Stability of the Biuret reagent
- 3 Procedure
- 3.1 Technique
- 3.2 Calibration curve
- 4 Interferences
- 4.1 Substances that interfere with the Biuret test
- 4.2 Substances that do not interfere with the Biuret test
- 5 Advantages
- 6 Disadvantages
- 7 Uses
- 7.1 Pathologies that occur with an increase or decrease in proteins
- 7.2 Clinical samples
- 7.3 Non-clinical samples
- 8 References
Basis
Biuret's reagent is composed of potassium hydroxide, cupric sulfate, and sodium and potassium tartrate. Sodium hydroxide is used to alkalize the medium, since this condition is essential for the reaction to take place.
The substances that react with proteins is cupric sulfate, while sodium tartrate has the function of not allowing the formation of copper hydroxide, which tends to precipitate and interferes with the reaction..
If substances with peptide bonds (polypeptides or proteins) are found in the sample, the test will be positive.
A reaction is interpreted as positive when the solution turns purple. The color is produced by the formation of a complex between at least two peptide bonds that have the CO-NH group and the cupric cations.
The violet complex can be formed in two ways: one is by the loss of protons from the amide groups that bind to the metal (depronation), and the other by the union of the oxygen and nitrogen electrons that are free and join with copper.
This reaction can vary in intensity and color depending on the type of protein..
The test can be performed qualitatively or quantitatively. In the qualitative form, it is reported as positive or negative. While in the quantitative form the concentration can be measured by the spectrophotometric method.
The reaction is read between 540-560 nm. The intensity of the color is directly proportional to the concentration of peptide bonds in the sample..
Reagents
-Sodium hydroxide (NaOH) 20%
-1% Cupric Sulfate Pentahydrate (CuSO4. 5HtwoOR)
-Mixed sodium potassium tartrate tetrahydrate (KNaC4H4OR64HtwoOR)
Biuret reagent stability
-It must be kept refrigerated.
Process
Technique
-Place 100 µl of the sample or standard to be analyzed in a test tube.
-Add 2 ml of sodium hydroxide.
-Mix very well.
-Add 5 ml of Biuret reagent.
-Mix and let stand 25 minutes at room temperature, cover and protect from light.
-Observe the formation or not of color and measure spectrophotometrically.
Calibration curve
Bovine serum albumin can be used as a standard to perform the calibration curve. Various concentrations are prepared from it. For example 25, 50, 75, 100, 125 and 150%.
The reaction is set up with all these known concentrations and the absorbance is read at a wavelength of 540 nm. With the data of the known concentrations and the absorbance readings the calibration curve is made.
In each determination or batch of processed samples it is recommended to mount a standard. Bovine serum albumin 0.1-2 mg / ml can be used as a calibration standard..
Measurements are made on a spectrophotometer at 540 nm.
Linearity is met up to a concentration of 12 g / dl.
Interference
Substances that interfere with the Biuret test
Although it is not very frequent, it should be noted that some substances may interfere during the execution of this test. For example, the presence of ammonia can inhibit the formation of color.
Likewise, other substances could absorb at the same wavelength, such as certain pigments..
On the other hand, interference can be generated when a substance other than the peptide bond forms a complex with the cupric salt. Example: some carbohydrates and certain lipids.
If the sample to be analyzed presents some type of precipitate, it must be filtered or centrifuged before mounting the test..
Substances that do not interfere with the Biuret test
The test is not affected by the presence of:
-Bilirubin up to a concentration of 20 mg / dl.
-Hemoglobin up to a concentration of 750 mg / dl.
-Dextran up to a concentration of 30 g / L.
-Triglycerides up to a concentration of 4000 mg / dl.
Advantage
-It is a simple method to execute.
-It is an economic test.
-Has high specificity for proteins.
-Little interference.
Disadvantages
It has poor sensitivity to detect low amounts of protein. The work carried out by Fuentes and collaborators affirms that the Biuret test method has a detection limit of 1 mg / ml of protein and a limit of quantification of 3 mg / ml.
However, other research conducted at the University of Amazonia report much lower values. The detection limit reported by the study is 0.020 mg / ml and the quantification limit is 1.33 mg / ml..
Applications
The Biuret reagent or test is used for the determination of proteins in clinical and non-clinical samples in routine and research laboratories.
Pathologies that occur with an increase or decrease in protein
In many pathologies it is important to determine the concentration of total proteins in clinical samples, which may be high or low.
They are elevated in:
-Multiple myeloma,
-Systemic lupus erythematosus,
-Bacterial endocarditis,
-Bacterial meningitis,
Waldenstrom's macroglobulinemia, among others.
It is decreased in:
-Renal insufficiency,
-People with severe degrees of malnutrition,
-Patients with chronic infections, among others.
Clinical samples
The most common clinical samples are serum, plasma, and urine. The normal value of proteins in serum or plasma is 6.0-8.8 gr / dl.
The concentration of protein in the urine in adults does not exceed the figure of 150 mg / 24 hours.
Urine protein / urine creatinine ratio normal value
Infants: < a 0,50 mg
Children 2 years and older: index: 0.20 mg
Adults: < 0,2 mg
Non-clinical samples
The Biuret reaction can be used for many types of non-clinical samples, such as dairy products, antivenom, or any unknown substance for which you want to investigate the presence of proteins..
References
- Vázquez J, Guerra L, Quintana J, Ramírez J, Fernando Ry Vázquez Y. (2014). Physico-chemical characterization and protein content of fluid extracts of the mangrove oyster (Crassostrearizophorae). Cuban Journal of Chemistry, 26 (1), 66-74. Retrieved on June 26, 2019, from http: //scielo.sld
- Chaparro S, Lara A, Sandoval A, Sosa S, Martínez J, Gil J. Functional characterization of almond from mango seeds (Mangifera indica L.) Science in Development Magazine. 2015; 6 (1): 67-75
- "Biuret." Wikipedia, The Free Encyclopedia. 19 Jun 2019, 16:37 UTC. 26 Jun 2019, 22:18
- Fuentes F, Quispe I, García J. Standardization of the Biuret method to quantify total proteins in polyvalent antibotropic serum produced at the National Center for Biological Products of the INS. Bol - Inst Nac Salud 2012; 18 (11-12). Available at: repositorio.ins.gob.pe
- Winer Laboratories. Total proteins. Colorimetric method for the determination of total proteins in serum and plasma. Available at: wiener-lab.com.ar



Yet No Comments