
Potassium carbonate (K2CO3) structure, properties, uses, production

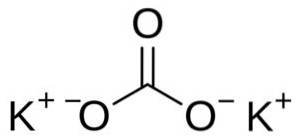
The Potassium carbonate is an inorganic compound formed by two potassium K ions+ and a carbonate ion CO3two-. Its chemical formula is KtwoCO3. It is a hygroscopic white solid, that is, it easily absorbs water from the environment. For this reason, in laboratories it is used to absorb water from other substances..
It is very soluble in water, forming alkaline solutions, which are rich in OH ions.-and therefore with a high pH value. Its aqueous solutions, being alkaline, are used in various industrial processes to absorb acid gases such as carbon dioxide COtwo and hydrogen sulfide HtwoYes, it easily neutralizes them.

The KtwoCO3 It is used to prepare soaps, cleaning products, laundry detergents, and dishwashing mixes. It is also used in the processing of some textile fibers such as wool..
It is widely used in chemistry laboratories, for example to absorb water from other compounds or to alkalize mixtures of chemical reactions and also in chemical analysis.
It is also added to some foods, for example, to eliminate the bitter taste of cocoa beans during the production of chocolate..
Article index
- 1 Structure
- 2 Nomenclature
- 3 Properties
- 3.1 Physical state
- 3.2 Molecular weight
- 3.3 Melting point
- 3.4 Boiling point
- 3.5 Density
- 3.6 Solubility
- 3.7 pH
- 3.8 Chemical properties
- 4 Obtaining
- 5 Uses
- 5.1 In the absorption of CO2 in industrial processes
- 5.2 In the removal of H2S from certain processes
- 5.3 In chemistry laboratories
- 5.4 In the cleaning products industry
- 5.5 In the food industry
- 5.6 In fertilizers
- 5.7 In various applications
- 6 References
Structure
Potassium carbonate is made up of two potassium K cations+ and a carbonate anion CO3two-. The carbonate anion has a flat and symmetrical structure, while the three oxygen atoms surround the carbon forming a flat triangle..
Nomenclature
- Potassium carbonate
- Potassium carbonate
- Dipotassium carbonate
- Potash
- Potassium salt of carbonic acid.
Properties
Physical state
Colorless to white crystalline solid.
Molecular weight
138.205 g / mol.
Melting point
899 ºC.
Boiling point
It decomposes.
Density
2.29 g / cm3
Solubility
Very soluble in water: 111 g / 100 g of water at 25 ° C. Insoluble in ethanol and acetone.
pH
An aqueous solution can have a pH of 11.6, that is, it is quite alkaline.
Chemical properties
Potassium carbonate is deliquescent or hygroscopic, that is, it absorbs moisture from the environment. It has a stable hydrate, KtwoCO3.2HtwoOR.
The KtwoCO3 in aqueous solution it hydrolyzes, that is, it reacts with water releasing OH groups- which are the ones that give alkalinity to the solutions:
CO3two- + HtwoO ⇔ OH- + HCO3-
HCO3- + HtwoO ⇔ OH- + HtwoCO3
Obtaining
It can be obtained from the ashes that remain when burning plants. Also by carbonation of potassium hydroxide KOH, that is, adding excess carbon dioxide COtwo to KOH:
KOH + COtwo → KHCO3
2 KHCO3 + heat → KtwoCO3 + HtwoOR
Another way to obtain it is by heating potassium chloride KCl with magnesium carbonate MgCO3, water and COtwo under pressure. A hydrated double salt of magnesium and potassium is obtained first MgCO3.KHCO3.4HtwoOr, called Engels salt:
2 KCl + 3 MgCO3 + COtwo + 5 htwoO → MgCO3.KHCO3.4HtwoO ↓ + MgCltwo
Engels' hydrated double salt precipitates and is filtered out of solution. Then it is heated and the potassium carbonate K is formedtwoCO3 that when adding water dissolves while magnesium carbonate MgCO3 remains insoluble and is removed by filtration.
MgCO3.KHCO3.4HtwoO + heat → MgCO3↓ + 2K+ + CO3two- + COtwo↑ + 9 HtwoOR
Applications
In the absorption of COtwo in industrial processes
The potassium carbonate solution is the classic treatment for the removal of carbon dioxide COtwo in various processes, especially in high pressure and high temperature applications.

CO removaltwo occurs according to the following reaction:
KtwoCO3 + COtwo + HtwoO ⇔ 2 KHCO3
This method is used for example to treat natural gas. Also in power generation plants, to avoid the emission of COtwo into the atmosphere, and in the production of dry ice.

K's solutiontwoCO3 can be thermally regenerated, that is, by heating to temperatures around 100 ° C.
So that the potassium carbonate solution can absorb the CO2 at a good speed, promoters are added that accelerate the process such as diethanolamine (DEA)..
In the removal of HtwoS of certain processes
Potassium carbonate solutions are also used to remove hydrogen sulfide gas HtwoS for process streams. Potassium K triphosphate is sometimes added3PO4 for make faster the process.
In chemistry laboratories
The KtwoCO3 It allows to carry out organic syntheses, for example, in condensation reactions and to neutralize. It is used to remove water from organic liquids, as a dehydrating agent or desiccant in the laboratory.
It is also used in analytical chemistry reactions and for alkalization in the pharmaceutical industry..
In the cleaning products industry
The KtwoCO3 It is used to make soap, cleaning formulas, laundry and dishwashing products, and also to prepare shampoo and other personal care products.

In the food industry
Potassium carbonate is added to various foods for various purposes.
For example, it is added to cocoa beans to remove their bitter taste and use them in the production of chocolate. It is added to the grapes in the drying process to obtain raisins.

In pastry it is used as a leavening agent (which acts as a yeast) for flour to prepare baked goods..

In fertilizers
The KtwoCO3 It is used to fertilize acidic soils, since the carbonate ion CO3two- in contact with water produces OH ions- that increase the pH of the soil. Plus potassium K+ it is a nutrient for plants.
Potassium carbonate has also been used to make slow release fertilizers.
A slow-release fertilizer releases or releases nutrients slowly so that they are not dissolved and washed away by the water. Thanks to this, they will be able to spend more time available to the roots of the plant.
In various applications
Potassium carbonate KtwoCO3 it is also used to:
- Raw wool dyeing, bleaching and cleaning processes and other activities of the textile industry
- Obtaining other organic and inorganic potassium salts, such as KCN potassium cyanide.
- Function as an acidity regulator in various processes.
- Making ceramics and pottery.
- Engraving and lithography processes.
- Tanning and finishing of leathers.
- Prepare inks for printing, pigments.
- Manufacture glasses especially for television, since the KtwoCO3 is more compatible than sodium carbonate NatwoCO3 with lead, barium and strontium oxides contained in these glasses.
- Water treatment.
- Retard fire (in the form of aqueous solutions).
- Inhibit corrosion and as an antifouling agent in process equipment.
References
- U.S. National Library of Medicine. (2019). Potassium carbonate. Recovered from pubchem.ncbi.nlm.nih.gov.
- Steele, D. (1966). The Chemistry of the Metallic Elements. Pergamon Press Ltd. London.
- Mokhatab, S. et al. (2019). Natural Gas Treating. The Potassium Carbonate Solution. In Handbook of Natural Gas Transmission and Processing (Fourth Edition). Recovered from sciencedirect.com.
- Kakaras, E. et al. (2012). Pressurized fluidized bed combustion (PFBC) combined cycle systems. Pressurized fluidized bed combustion with carbon capture and storage. In Combined Cycle Systems for Near-Zero Emission Power Generation. Recovered from sciencedirect.com.
- Speight, J.G. (2019). Hydrogen Production. Wet Scrubbing. In Heavy Oil Recovery and Upgrading. Recovered from sciencedirect.com.
- Branan, C.R. (2005). Gas treating: Chapter updated by Chris Higman. Hot Carbonate Processes. In Rules of Thumb for Chemical Engineers (Fourth Edition). Recovered from sciencedirect.com.
- Kirk-Othmer (1994). Encyclopedia of Chemical Technology. Fourth Edition. John Wiley & Sons.
- Ullmann's Encyclopedia of Industrial Chemistry. (1990). Fifth Edition. VCH Verlagsgesellschaft mbH.
- Li, Y. and Cheng, F. (2016). Synthesis of a novel slow-release potassium fertilizer from modified Pidgeon magnesium slag by potassium carbonate. J Air Waste Manag Assoc, 2016 Aug; 66 (8): 758-67. Recovered from ncbi.nlm.nih.gov.

Yet No Comments