
Lauric acid structure, properties, uses, benefits, effects
The lauric acid It is a medium-chain, 12-carbon saturated fatty acid. It consists of a white solid or bright white powder, with a characteristic baby oil smell. It is practically insoluble in water, but it is very soluble in organic solvents; especially in ethanol, methanol and acetone.
It is one of the components of triglycerides. Coconut milk is very rich in this fatty acid (around 50% of the fatty acids). It is also found in coconut oil and palm kernel oil. In breast milk, it represents 6.2% of total lipids.

Lauric acid is metabolized to give rise to monolaurin, a compound to which activity against great-negative bacteria, viruses, fungi and protozoa is attributed..
Lauric acid has been shown to be helpful in treating acne, as it has activity against bacteria Propionibacterium acnes, related to the inflammatory processes seen in the disorder.
Likewise, it has been used experimentally in colon rectal cancer cell cultures, being capable of inducing apoptosis of these cells; which could open the door to the use of lauric acid in an alternative therapy for this type of cancer
In summary, lauric acid has numerous health benefits, also being used in the preparation of cosmetics, fragrances, soaps, shampoos, etc..
Article index
- 1 Structure of lauric acid
- 2 Properties
- 2.1 Chemical names
- 2.2 Molecular formula
- 2.3 Molecular weight
- 2.4 Physical description
- 2.5 Odor
- 2.6 Melting point
- 2.7 Boiling point
- 2.8 Solubility in water
- 2.9 Solubility in organic solvents
- 2.10 Density
- 2.11 Vapor pressure
- 2.12 Viscosity
- 2.13 Heat of combustion
- 2.14 Surface tension
- 2.15 Refractive index
- 2.16 Dissociation constant
- 3 Uses
- 3.1 Industrial
- 3.2 Determination of molar mass
- 3.3 Agriculture
- 4 Health benefits
- 4.1 Vascular bypass grafts
- 4.2 Increases the action of certain drugs
- 4.3 Bactericidal action
- 4.4 Treatment of the rectal colon
- 4.5 Fight infections and pathogens
- 4.6 Others
- 5 Side effects
- 6 References
Structure of lauric acid
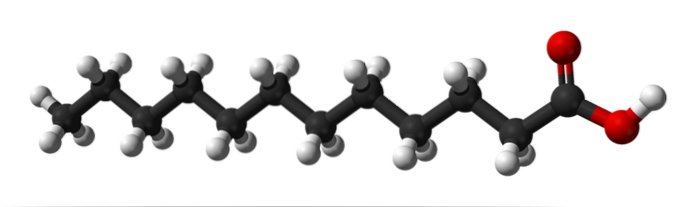
The upper image shows the molecular structure of lauric acid using a spheres and bars model..
It can be counted that in its carbon "tail" there are eleven carbon atoms or black spheres; it constitutes the apolar or hydrophobic end of the structure. And to its right, the polar head with the COOH group, with its red spheres. There are twelve carbon atoms in all.
The COOH group can donate an H ion+, forming salts of laurates. In these, the hydrogen of the OH group, located at the end of the right, is replaced by metal cations.
The apolar tail of lauric acid is free of unsaturations. What does it mean? Lacking double bonds (C = C). If it did, it would have folds, characteristic of unsaturated fats. All carbons are bound to the maximum number of hydrogen allowed, and that is why this fat is of the saturated type.
Intermolecular interactions
The structure in general presents a too obvious form: that of a zigzag. Not only that, it is also a flexible zigzag, capable of arranging its carbon atoms in such a way that they perfectly match the empty spaces in the tail of a neighboring molecule..
A good analogy to understand this is to compare the interactions between two lauric acid molecules with that of two folding chairs that are stacked on top of each other to form a column; The same happens with these molecules: their saturated nature allows them to group together and precipitate into a fat of greater insolubility.
The apolar tails therefore interact by means of London scattering forces. Meanwhile, the polar heads form hydrogen bonds between them, which further strengthens the bonds of their tails..
Thus, the molecules are coupled in an orderly fashion until they form a white crystal with a triclinic structure..
These greasy lauric acid crystals are not very resistant to heat, since they melt at 43 ° C. The increase in temperature uncouples the tails as a result of the vibrations of their atoms; they begin to slide over each other, thus giving rise to lauric acid oil.
Properties
Chemical names
Lauric acid, n-dodecanoic acid, dodecylic acid, dodecanoic acid, volvic acid and dodecylcarboxylate.
Molecular formula
C12H24ORtwo and CH3(CHtwo)10COOH.
Molecular weight
200.322 g / mol
Physical description
Solid white. It can also present as colorless needles or a white crystalline powder..
Odor
Characteristic of baby oil.
Melting point
43.2 ºC (111 ºF).
Boiling point
297.9 ºC
Water solubility
Practically insoluble in water:
-37 mg / L at 0 ºC
-48.1 mg / L at 50 ºC
-83 mg / L at 100 ºC.
Solubility in organic solvents
One gram of lauric acid is dissolved in 1 mL of ethyl alcohol and 2.5 mL of propyl alcohol. It is highly soluble in benzene and ether, but slightly soluble in chloroform.
Solubility in acetone
60.5 g / 100 g at 20 ºC
Solubility in methanol
120 g / 100 g at 20 ºC.
Density
-1.007 g / cm3 at 24 ºC
-0.8744 g / cm3 at 41.5 ºC
-0.8679 g / cm3 at 50 ºC.
Vapor pressure
-2.13 x 10-6 kPa (25 ºC)
-0.42 kPa (150 ºC).
Viscosity
-6.88 cPoise (50 ºC)
-5.37 cPoise (60 ºC).
Heat of combustion
7,413.7 kJ / mol.
Surface tension
26.6 mN / m at 70 ºC.
Refractive index
1.4183 at 82 ºC.
Dissociation constant
pKa = 5.3 at 20 ºC.
Applications
Industrial
Lauric acid reacts with sodium hydroxide to saponify it, producing sodium laurate. Soaps made with lauric acid are foamy and have the ability to dissolve lipids.
Fractionated fatty acids, including lauric acid, are used in the manufacture of esters, fatty alcohols, peroxides, fragrances, surface finishing, lubricants, cosmetics, animal feed, paper chemistry, plastics, detergents, agrochemicals, resins and coatings.
Determination of molar mass
Lauric acid is used in determining the unknown molar mass of a substance. The reason: it has a relatively high melting point (43.8ºC). Lauric acid and the other substance are melted, determining the decrease in the cryoscopic point of the mixture and thus the molar mass of the substance..
farming
Lauric acid is used in agriculture as a herbicide, insecticide, miticide, and plant growth regulator. Mosses, algae, lichens, liverworts, and other weeds are also used in feed crops, ornamental flowers, and household plants..
Health benefits
Vascular bypass grafts
Lauric acid binds to the peptide gli-arg-gli-asp-ser (GRGDS), facilitating its incorporation into the matrix of poly (carbonate-urea) urethane (PCU), used in grafts for vascular bypass.
Increases the action of certain drugs
It facilitates the transdermal penetration of the drug fenazepam, increasing the anticonvulsant action of the drug three times. It also increases the passage through moist skin of the drugs neutral caffeine and anionic sodium salicylate..
Bactericidal action
In an investigation, the bactericidal action of lauric acid was studied, both in vivo and in vitro, on the bacteria Staphylococcus aureus, Staphylococcus epidermis and Propionabacterium acnes, bacteria that cause vascular inflammation..
A dose 15 times lower of lauric acid was required, compared to the dose of benzoyl peroxide (BPO), a drug usually used in the treatment of acne, to show a therapeutic effect.
Of the bacteria studied, P. acnes was the most sensitive to lauric acid. Furthermore, lauric acid did not show a cytotoxic effect on human sebocytes..
Rectal colon treatment
Lauric acid is being studied as a promising adjunct therapy in the treatment of colon rectal cancer. Noting that lauric acid reduces the use of glutathione (GSH), and produces an oxidative stress that induces apoptosis of colon rectal cancer cells.
Fight infections and pathogens
-Lauric acid is used to treat viral infections, including influenza and genital herpes caused by the herpes simplex virus (HSV). Additionally, lauric acid is used to treat genital warts caused by papillomavirus (HPV)..
-Serves as a precursor for monolaurin, an antimicrobial substance that fights pathogens such as bacteria, viruses and fungi.
-By itself or in conjunction with essential oils, it can combat pathogens present in food. Also, when fed to farm animals, it can improve the quality of the food produced by them..
Others
-It could be beneficial in the treatment of dry skin, as it could help in its hydration. This property could be used to improve diseases such as psoriasis.
It has an antioxidant property. Increases the levels of high-density lipoprotein (HDL), which works by removing cholesterol deposited in blood vessels, allowing its exit from the body.
-Lowers blood pressure and heart rate. In addition, it reduces oxidative stress present in the heart and kidneys..
Side effects
Lauric acid is a saturated fatty acid, so it is thought that it can cause damage to the cardiovascular system; since saturated fats cause a deposit of cholesterol in the blood vessels.
However, according to a 2016 review, the damaging action of lauric acid on the functioning of the cardiovascular system is not conclusive. A similar conclusion was reached in a review carried out in 2003.
As lauric acid is a carboxylic acid, a set of non-specific side effects can be attributed to it. For example, reacting with diazo, dithiocarbamate, isocyanate, mercaptan, nitrides, and sulfides can produce toxic gases.
However, these are not specific side effects of lauric acid, nor has it been shown to be involved in these reactions. In general, no side effects have been found..
But, some harmful effects that can occur during handling are known. May be harmful by inhalation, ingestion, or direct skin contact. In vapor form, it is irritating to the eyes, mucous membrane, nose and respiratory tract..
References
- Nakatsuji, T., Kao, M. C., Fang, J. Y., Zouboulis, C. C. (2009). Antimicrobial Property of Lauric Acid Against Propionibacterium Acnes: Its Therapeutic Potential for Inflammatory Acne Vulgaris. Journal Investigative Dermatology 129 (10): 2480-2488.
- Fauser, J. K., Matthews, G. S., Cummins, A. and Howarth, G. (2013). Induction of Apoptosis by the Medium-chain Length Fatty Acid Lauric in Colon Cancer Cells due to Induction of Oxidative Stress. Chemotheraphy 59 (3): 214-224.
- Superfoodly. (October 06, 2017). 25 Lauric Acid Foods & Oils: High Sources To Supplement With. Recovered from: superfoodly.com
- Tuscany Diet. (2019). Lauric acid: structure, properties and sources. Recovered from: tuscany-diet.net
- R. Lomer. (1963). The crystal and molecular structure of lauric acid (form A). Cryst Act. 16, 984.
- National Center for Biotechnology Information. (2019). Lauric acid. PubChem Database. Recovered from: pubchem.ncbi.nlm.nih.gov
- WebMD LLC. (2019). Lauric acid. Recovered from: webmd.com
- Schaefer Anna. (February 22, 2019). What is lauric acid? Recovered from: healthline.com
- Lama C. Siddhi. (April 12, 2019). Lauric Acid's Benefits for the Body. Recovered from: livestrong.com



Yet No Comments