
Aufbau Principle Concept and Explanation, Examples
The Aufbau principle It consists of a useful guide to theoretically predict the electronic configuration of an element. The word aufbau refers to the German verb "to build." The rules dictated by this principle are intended to "help build the atom".
When talking about the hypothetical atomic construction, it refers exclusively to electrons, which in turn go hand in hand with the increasing number of protons. Protons define the atomic number Z of a chemical element, and for each one added to the nucleus an electron is added to compensate for this increase in positive charge..
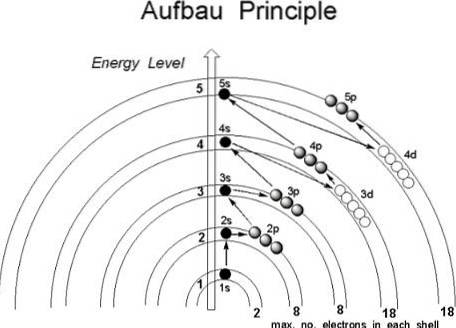
Although it seems that the protons do not follow an established order to join the nucleus of the atom, the electrons do follow a series of conditions, in such a way that they first occupy the regions of the atom with lower energy, specifically those where the probability of finding them in space is greater: the orbitals.
The Aufbau principle, along with other electronic filling rules (the Pauli exclusion principle and Hund's rule), helps to establish the order in which electrons should be added to the electron cloud; in this way, it is possible to assign a proper electronic configuration of a certain chemical element.
Article index
- 1 Concept and explanation
- 1.1 Layers and sublayers
- 1.2 Pauli's exclusion principle and Hund's rule
- 2 Examples
- 2.1 Carbon
- 2.2 Oxygen
- 2.3 Calcium
- 3 Limitations of the Aufbau principle
- 4 References
Concept and explanation
If the atom were considered as if it were an onion, a finite number of layers would be found within it, determined by the principal quantum number n.
Further, inside them, are the subshells, whose shapes depend on the quantum numbers azimuthal l and magnetic m.
The orbitals are identified by the first three quantum numbers, while the fourth, the spin s, ends up indicating in which orbital the electron will be located. It is then in these regions of the atom where the electrons rotate, from the innermost to the outermost layers: the valence layer, the most energetic of all..
This being the case, in what order should the electrons fill the orbitals? According to the Aufbau principle, they should be assigned based on the increasing value (n + l).
Likewise, within the subshells (n + l) the electrons must occupy the subshell with the lowest energy value; in other words, they occupy the lowest value of n.
Following these construction rules, Madelung developed a visual method that consists of drawing diagonal arrows, which help to build the electronic configuration of an atom. In some educational spheres this method is also known as the rain method..
Layers and sublayers
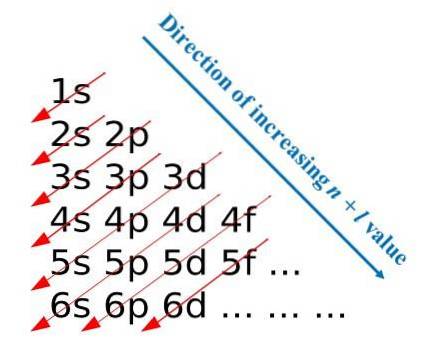
The first image illustrates a graphical method to obtain the electron configurations, while the second image is the respective Madelung method. The most energetic layers are located at the top and the least energetic are in a downward direction..
From left to right, the sub-layers s, p, d and f of their corresponding main energy levels are “transited”. How to calculate the value of (n + l) for each step marked by the diagonal arrows? For example, for the 1s orbital this calculation is equal to (1 + 0 = 1), for the 2s orbital (2 + 0 = 2), and for the 3p orbital (3 + 1 = 4).
The result of these calculations originates the construction of the image. Therefore, if it is not available at hand, it is enough to determine (n + l) for each orbital, starting to fill the orbitals with electrons from the one with the smallest value of (n + l) to the one with the maximum value..
However, using the Madelung method greatly facilitates the construction of the electron configuration and makes it an entertaining activity for those who are learning the periodic table..
Pauli's exclusion principle and Hund's rule
Madelung's method does not indicate the orbitals of the subshells. Taking them into account, the Pauli exclusion principle states that no electron can have the same quantum numbers as another; or what is the same, a pair of electrons cannot have both positive or negative spins.
This means that their spin quantum numbers s cannot be equal and, therefore, their spins must pair when occupying the same orbital.
On the other hand, the filling of the orbitals must be done in such a way that they are degenerate in energy (Hund's rule). This is achieved by keeping all the electrons in the orbitals unpaired, until it is strictly necessary to pair a pair of these (as with oxygen).
Examples
The following examples summarize the whole concept of the Aufbau principle..
Carbon
To determine its electronic configuration, the atomic number Z must first be known, and thus, the number of electrons. Carbon has Z = 6, so its 6 electrons must be located in the orbitals using the Madelung method:

The arrows correspond to the electrons. After filling the 1s and 2s orbitals, each with two electrons, the 2p orbitals by difference are assigned the remaining two electrons. Hund's rule is thus manifested: two degenerate orbitals and one empty.
Oxygen

Oxygen has Z = 8, so it has two extra electrons unlike carbon. One of these electrons must be placed in the empty 2p orbital, and the other must pair to form the first pair, with the arrow pointing downward. Consequently, here the Pauli exclusion principle is manifested.
Calcium
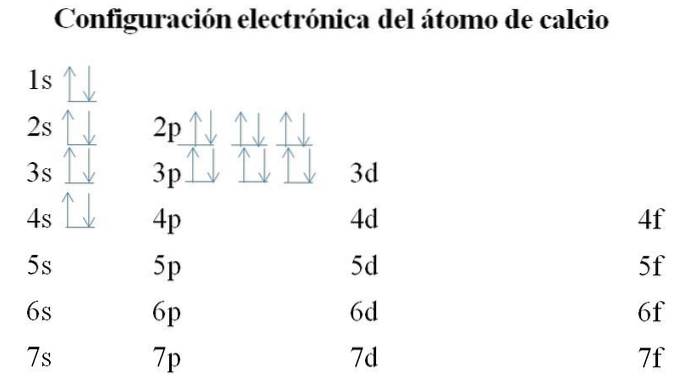
Calcium has 20 electrons, and the orbitals are still filled with the same method. The order of filling is as follows: 1s-2s-2p-3s-3p-4s.
It can be noted that instead of filling the 3d orbital first, the electrons occupy the 4s. This occurs before making way for transition metals, elements that do fill the inner layer 3d.
Limitations of the Aufbau principle
Aufbau's principle fails to predict the electronic configurations of many transition metals and rare earth elements (lanthanides and actinides).
This is because the energetic differences between the ns and (n-1) d orbitals are low. For reasons supported by quantum mechanics, electrons may prefer to degenerate the (n-1) d orbitals at the cost of undoing or dislodging the electrons from the ns orbital..
A famous example is the case of copper. Its electron configuration predicted by the Aufbau principle is 1stwo2stwo2 P63stwo3p64stwo3d9, when experimentally it has been shown to be 1stwo2stwo2 P63stwo3p64s13d10.
In the first, a solitary electron is unpaired in a 3d orbital, while in the second, all the electrons in the 3d orbitals are paired..
References
- Helmenstine, Anne Marie, Ph.D. (June 15, 2017). Aufbau Principle Definition. Taken from: thoughtco.com
- Prof. N. De Leon. (2001). The Aufbau Principle. Taken from: iun.edu
- Chemistry 301. Aufbau Principle. Taken from: ch301.cm.utexas.edu
- Hozefa Arsiwala and teacherlookup.com. (June 1, 2017). In Depth: Aufbau Principle With Examples. Taken from: teacherlookup.com
- Whitten, Davis, Peck & Stanley. Chemistry. (8th ed.). CENGAGE Learning, p 199-203.
- Goodphy. (July 27, 2016). Scheme of Madelung. [Figure]. Taken from: commons.wikimedia.org



Yet No Comments