
What is a saturated solution? (with examples)

A saturated solution It is a chemical solution that contains the maximum concentration of solute dissolved in a solvent. It is considered a state of dynamic equilibrium where the speeds at which the solvent dissolves the solute and the recrystallization speed are equal (J., 2014).
The additional solute will not dissolve in a saturated solution and will appear in a different phase, either a precipitate if it is a solid in liquid or an effervescence if it is a gas in a liquid (Anne Marie Helmenstine, 2016).
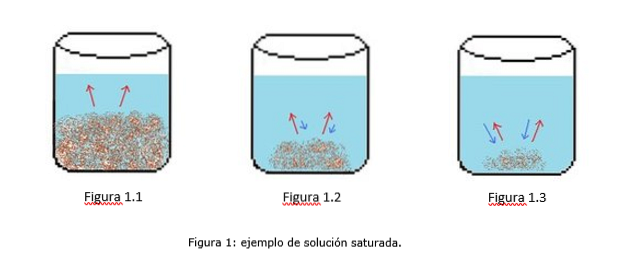
An example of a saturated solution is illustrated in Figure 1. In Figures 1.1, 1.2, and 1.3, there is a constant volume of water in the beaker. In figure 1.1 the saturation process begins, where the solute begins to dissolve, represented by the red arrows.
In figure 1.2, much of the solid has dissolved, but not completely due to the recrystallization process, represented by the blue arrows..
In Figure 1.3, only a small amount of solute remains undissolved. In this case, the recrystallization rate is greater than the dissolution rate. (tipes of saturation, 2014)
The point of maximum concentration of a solute in a solvent is known as the saturation point.
Article index
- 1 Factors affecting saturation
- 1.1 Temperature
- 1.2 Pressure
- 1.3 Chemical composition
- 1.4 Mechanical factors
- 2 Saturation and solubility curves
- 3 Examples of saturated solutions
- 4 What is a supersaturated solution?
- 5 References
Factors affecting saturation
The amount of solute that can be dissolved in a solvent will depend on different factors, among them the most important are:
Temperature
Solubility increases with temperature. For example, more salt can be dissolved in hot water than in cold water..
However, there may be exceptions, for example, the solubility of gases in water decreases with increasing temperature. In this case, the solute molecules receive kinetic energy as they heat up, which facilitates their escape..
Pressure
Increased pressure can force solute dissolution. This is commonly used to dissolve gases in liquids..
Chemical composition
The nature of the solute and solvent and the presence of other chemicals in the solution affect solubility. For example, more sugar can be dissolved in water than salt in water. In this case it is said that sugar is more soluble.
Ethanol in water are completely soluble with each other. In this particular case, the solvent will be the compound found in greater quantity.
Mechanical factors
In contrast to the dissolution rate, which depends mainly on temperature, the recrystallization rate depends on the concentration of solute on the surface of the crystal lattice, which is favored when a solution is immobile..
Therefore, stirring the solution avoids this accumulation, maximizing dissolution (tipes of saturation, 2014).
Saturation and solubility curves
The solubility curves are a graphical database where the amount of solute that dissolves in a quantity of solvent is compared, at a certain temperature.
Solubility curves are commonly plotted for a quantity of solute, either solid or gas, in 100 grams of water (Brian, 2014).
Figure 2 illustrates the saturation curves for various solutes in water..
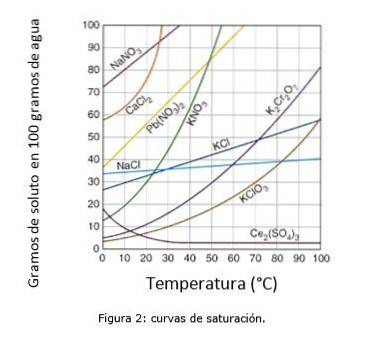
On the axis of the coordinates we have the temperature in degrees centigrade, and on the axis of the abscissa we have the concentration of solute expressed in grams of solute per 100 grams of water.
The curve indicates the saturation point at a given temperature. The area under the curve indicates that you have an unsaturated solution and therefore more solute can be added.
The area above the curve has a supersaturated solution. (Solubility Curves, n.d.)
Taking sodium chloride (NaCl) as an example, at 25 degrees centigrade approximately 35 grams of NaCl can be dissolved in 100 grams of water to obtain a saturated solution. (Cambrige University, s.f.)
Examples of saturated solutions
Saturated solutions can be found on a day-to-day basis, it is not necessary to be in a chemical laboratory. The solvent does not necessarily have to be water. Below are everyday examples of saturated solutions:
-Soda and soft drinks are generally saturated solutions of carbon dioxide in water. That is why, when the pressure is released, bubbles of carbon dioxide form..
-Earth soils are saturated with nitrogen.
-Sugar or salt can be added to the vinegar to form a saturated solution..
-Add powdered chocolate to milk until it does not dissolve, it forms a saturated solution.
-Milk can be saturated with flour to such an extent that no more flour can be added to the milk.
-Melted butter can be saturated with salt, when the salt no longer dissolves.
What is a supersaturated solution?
The definition of a supersaturated solution is one that contains more dissolved solute than could normally be dissolved in the solvent. This is usually done by increasing the temperature of the solution..
A slight alteration of the solution or introduction of a “seed” or tiny solute crystal will force crystallization of the excess solute. If there is no nucleation point for crystal formation, excess solute can remain in solution.
Another form of supersaturation can occur when a saturated solution is carefully cooled. This change in conditions means that the concentration is actually higher than the saturation point, the solution has been supersaturated..
This can be used in the recrystallization process to purify a chemical: it dissolves to saturation point in hot solvent, then as the solvent cools and the solubility decreases, excess solute precipitates.
Impurities, which are present in a much lower concentration, do not saturate the solvent and thus remain dissolved in the liquid.
References
- Anne Marie Helmenstine, P. (2016, July 7). Saturated Solution Definition and Examples. Retrieved from about: about.com
- Cambrige University. (s.f.). Solubility curves. Retrieved from dynamicscience.com: dynamicscience.com.au.
- Examples of Saturated Solution. (s.f.). Retrieved from yourdcitionary: examples.yourdictionary.com.
- , S. (2014, June 4). Saturated and Supersaturated Solutions. Retrieved from socratic.org: socratic.org.
- James, N. (s.f.). Saturated Solution: Definition & Examples. Retrieved from study.com: study.com.
- , B. (2014, October 14). Saturated and Supersaturated Solutions. Retrieved from socratic.org: socratic.org.
- Solubility Curves. (s.f.). Retrieved from KentChemistry: kentchemistry.com.
- Tipes of saturation. (2014, June 26). Retrieved from chemistry libretex: chem.libretexts.org.



Yet No Comments