
Plastoquinone classification, chemical structure and functions
The plastoquinone (PQ) is an organic lipid molecule, specifically an isoprenoid of the quinone family. In fact, it is a side chain polyunsaturated derivative of quinone that participates in photosystem II of photosynthesis..
Located in the thylakoid membrane of chloroplasts, it is apolar in character and is very active at the molecular level. Indeed, the name of plastoquinone derives from its location in the chloroplasts of higher plants.
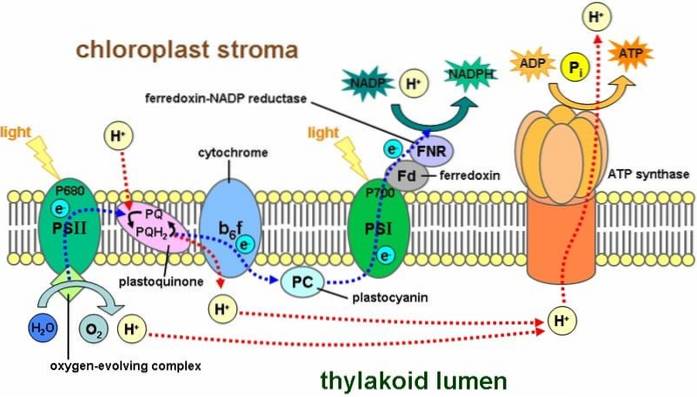
During photosynthesis, solar radiation is captured in the FS-II system by chlorophyll P-680 and then oxidized by releasing an electron. This electron rises to a higher energy level, which is picked up by the voter acceptor molecule: plastoquinone (PQ)..
Plastoquinones are part of the photosynthetic electron transport chain. They are the site of integration of different signals and a key piece in RSp31's response to light. There are about 10 PQs per FS-II that are reduced and oxidized according to the functional state of the photosynthetic apparatus..
Therefore, the electrons are transferred through a transport chain in which several cytochromes are involved, to later reach plastocyanin (PC), which will give the electrons to chlorophyll molecules of FS-I.
Article index
- 1 Classification
- 2 Chemical structure
- 2.1 -Biosynthesis
- 3 Functions
- 3.1 Light phase (PS-II)
- 4 References
Classification
Plastoquinone (C55H80ORtwo) is a molecule associated with a benzene ring (quinone). Specifically, it is an isomer of cyclohexadione, characterized by being an aromatic compound differentiated by its redox potential.
Quinones are grouped based on their structure and properties. Within this group, benzoquinones are differentiated, generated by the oxygenation of hydroquinones. The isomers of this molecule are the ortho-benzoquinone and in order to-benzoquinone.
On the other hand, plastoquinone is similar to ubiquinone, because they belong to the benzoquinone family. In this case, both serve as electron acceptors in transport chains during photosynthesis and anaerobic respiration..
Associated with its lipid status, it is categorized in the terpene family. That is, those lipids that make up plant and animal pigments, providing color to cells.
Chemical structure
Plastoquinone is made up of an active benzene-quinone ring associated with a side chain of a polyisoprenoid. In fact, the hexagonal aromatic ring is linked to two oxygen molecules through double bonds at carbons C-1 and C-4..
This element has the side chain and is composed of nine isoprenes linked to each other. Consequently, it is a polyterpene or isoprenoid, that is, hydrocarbon polymers of five carbon atoms isoprene (2-methyl-1,3-butadiene).
Likewise, it is a prenylated molecule, which facilitates attachment to cell membranes, similar to lipid anchors. In this regard, a hydrophobic group has been added to its alkyl chain (methyl group CH3 branched in position R3 and R4).
-Biosynthesis
During the photosynthetic process, plastoquinone is continuously synthesized, due to its short life cycle. Studies in plant cells have determined that this molecule remains active between 15 to 30 hours.
Indeed, plastoquinone biosynthesis is a very complex process, involving up to 35 enzymes. Biosynthesis has two phases: the first occurs in the benzene ring and the second in the side chains.
Initial phase
In the initial phase, the synthesis of the quinone-benzene ring and the prenyl chain is carried out. The ring obtained from tyrosines and prenyl side chains are the result of glyceraldehyde-3-phosphate and pyruvate..
Based on the size of the polyisoprenoid chain, the type of plastoquinone is established.
Ring condensation reaction with side chains
The next phase comprises the condensation reaction of the ring with the side chains.
Homogentistic acid (HGA) is the predecessor of the benzene-quinone ring, which is synthesized from tyrosine, a process that occurs thanks to the catalysis of the enzyme tyrosine amino-transferase.
For their part, the prenyl side chains originate in the methyl-erythritol phosphate (MEP) pathway. These chains are catalyzed by the enzyme solanesyl diphosphate synthetase to form solanesyl diphosphate (SPP).
Methyl erythritol phosphate (MEP) constitutes a metabolic pathway for the biosynthesis of Isoprenoids. After the formation of both compounds, the condensation of homogenistic acid occurs with the solanesyl diphosphate chain, a reaction catalyzed by the enzyme homogentistate solanesyl-transferase (HST).
2-dimethyl-plastoquinone
Finally, a compound called 2-dimethyl-plastoquinone is originated, which later with the intervention of the enzyme methyl-transferase, allows to obtain as a final product: plastoquinone.
Features
Plastoquinones intervene in photosynthesis, a process that occurs with the intervention of energy from sunlight, resulting in energy-rich organic matter from the transformation of an inorganic substrate.
Light phase (PS-II)
The function of plastoquinone is associated with the light phase (PS-II) of the photosynthetic process. The plastoquinone molecules involved in electron transfer are called Q A and Q B.
In this regard, photosystem II (PS-II) is a complex called water-plastoquinone oxido-reductase, where two fundamental processes are carried out. Water oxidation is enzymatically catalyzed and plastoquinone reduction occurs. In this activity, photons with a wavelength of 680 nm are absorbed.
Molecules Q A and Q B differ in the way they transfer electrons and the speed of the transfer. Also, due to the type of binding (binding site) with photosystem II. Q A is said to be the fixed plastoquinone and Q B is the mobile plastoquinone.
After all, Q A is the photosystem II binding zone that accepts the two electrons in a time variation between 200 and 600 us. Instead, Q B has the ability to bind and detach from photosystem II, accepting and transferring electrons to the cytochrome..
At the molecular level, when Q B is reduced, it is exchanged for another of the set of free plastoquinones within the thylakoid membrane. Between Q A and Q B there is a nonionic Fe atom (Fe+two) that participates in electronic transportation between them.
In summary, Q B interacts with amino acid residues in the reaction center. In this way, Q A and Q B acquire a large differential in the redox potentials..
Furthermore, since Q B is more loosely bound to the membrane, it can be easily separated by reducing to QH 2. In this state it is capable of transferring high-energy electrons received from Q A to cytochrome bc1-complex 8.
References
- González, Carlos (2015) Photosynthesis. Recovered at: botanica.cnba.uba.ar
- Pérez-Urria Carril, Elena (2009) Photosynthesis: Basic Aspects. Reduca (Biology). Plant Physiology Series. 2 (3): 1-47. ISSN: 1989-3620
- Petrillo, Ezequiel (2011) Regulation of alternative splicing in plants. Effects of light by retrograde signals and PRMT5 protein methyltransferase.
- Sotelo Ailin (2014) Photosynthesis. Faculty of Exact, Natural Sciences and Surveying. Chair of Plant Physiology (Study Guide).



Yet No Comments